Polivy and Yescarta have cleared South Korea’s Drug Reimbursement Evaluation Committee, expanding access to advanced Diffuse Large B-Cell Lymphoma (DLBCL) treatments. These therapies shift the clinical standard from the 20-year-old R-CHOP regimen toward targeted antibody-drug conjugates and CAR-T cell therapies for both first-line and relapsed patients.
For two decades, the R-CHOP regimen (a combination of rituximab and four chemotherapy agents) served as the undisputed ceiling for DLBCL care. While effective for many, a significant subset of patients experienced relapse or were refractory—meaning the cancer did not respond to treatment. The recent regulatory movement in Seoul reflects a global transition toward precision oncology, where the goal is to increase the “progression-free survival” (the length of time during and after treatment that a patient lives with the disease but it does not get worse) by targeting specific molecular markers.
In Plain English: The Clinical Takeaway
- Polivy acts like a “smart bomb,” delivering powerful chemotherapy directly to cancer cells while sparing more healthy tissue.
- Yescarta is a living therapy that reprogramms your own immune cells in a lab to hunt and destroy lymphoma cells.
- Insurance Coverage means these previously prohibitively expensive treatments are now accessible to a broader population of patients, not just those with extreme wealth.
The Molecular Shift: From Broad Chemotherapy to Targeted ADCs
The integration of Polatuzumab vedotin (Polivy) into first-line treatment marks a departure from traditional cytotoxic chemotherapy. Polivy is an antibody-drug conjugate (ADC). Its mechanism of action—the specific biochemical process through which a drug produces its effect—involves binding to the CD79b protein expressed on the surface of B-cells. Once bound, the drug is internalized by the cell, releasing a potent microtubule disruptor that triggers cell death.
Clinical data from large-scale, double-blind placebo-controlled trials—studies where neither the patient nor the doctor knows who is receiving the active drug to prevent bias—demonstrate that combining Polivy with R-CHP significantly improves outcomes compared to R-CHOP alone. By removing the vincristine (the ‘V’ in CHOP) and adding the targeted ADC, clinicians can reduce certain systemic toxicities while increasing the precision of the attack.
“The transition to ADC-based regimens in the first-line setting for DLBCL is not merely an incremental improvement; it is a fundamental change in how we approach aggressive B-cell malignancies, prioritizing molecular targeting over systemic toxicity,” notes a lead investigator in recent hematology literature.
CAR-T Cell Therapy: Reprogramming the Immune System
For patients who relapse after initial therapy, Axicabtagene ciloleucel (Yescarta) represents the cutting edge of immunotherapy. Unlike traditional drugs, Yescarta is a Chimeric Antigen Receptor (CAR) T-cell therapy. The process involves extracting a patient’s T-cells via leukapheresis, genetically modifying them to express a receptor that recognizes the CD19 antigen on lymphoma cells, and re-infusing them into the patient.
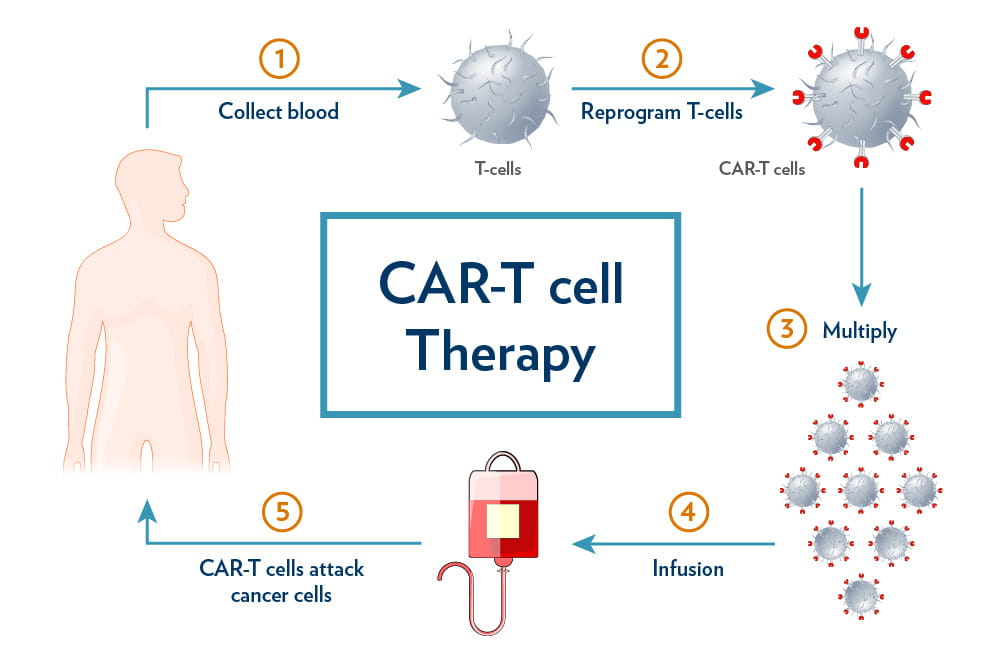
This “living drug” approach allows the immune system to maintain a surveillance presence in the body, potentially providing long-term remission for patients who previously had no viable options. However, this potency comes with a unique risk profile, most notably Cytokine Release Syndrome (CRS), an overactivation of the immune system that requires intensive monitoring in a clinical setting.
| Therapy | Drug Class | Primary Target | Typical Clinical Application | Key Advantage |
|---|---|---|---|---|
| R-CHOP | Chemotherapy/mAb | CD20 / DNA | Standard 1st Line | Established baseline efficacy |
| Polivy | ADC | CD79b | 1st Line & Relapsed | Reduced systemic toxicity |
| Yescarta | CAR-T Cell | CD19 | Relapsed/Refractory | Potential for long-term cure |
Global Access and the Reimbursement Gap
While the FDA in the United States and the EMA in Europe approved these agents years ago, the “Information Gap” for patients often lies in the difference between regulatory approval and reimbursement. In many healthcare systems, such as the NHS in the UK or the national insurance system in South Korea, a drug can be “approved” but remain inaccessible because the government has not agreed to pay for it based on cost-effectiveness ratios.
The decision by the Drug Reimbursement Evaluation Committee to move these drugs forward is a critical public health victory. It bridges the gap between clinical innovation and patient bedside. This shift is heavily influenced by the global trend of “value-based pricing,” where the high upfront cost of a CAR-T therapy like Yescarta is weighed against the long-term cost of repeated failed chemotherapy cycles and hospitalizations.
these trials were primarily funded by the manufacturers—Genmab and ADC Therapeutics for Polivy, and Kite Pharma (a Gilead company) for Yescarta. While the data is peer-reviewed and published in high-impact journals, the funding source underscores the need for independent, longitudinal studies to verify long-term survival rates beyond the initial trial windows.
Contraindications & When to Consult a Doctor
These advanced therapies are not suitable for all patients. Contraindications—specific situations in which a drug should not be used because it may be harmful—include:
- Severe Heart Failure: Patients with unstable cardiac function may not tolerate the systemic stress of CAR-T induction.
- Active Severe Infections: Because these therapies suppress or radically alter the immune system, active sepsis or uncontrolled infections are absolute barriers to treatment.
- Severe Neuropathy: Polivy can exacerbate peripheral neuropathy (nerve damage in the extremities); patients with pre-existing severe nerve damage must be evaluated carefully.
Patients should seek immediate medical intervention if they experience high fever, shortness of breath, or sudden confusion following an infusion, as these are hallmark signs of Cytokine Release Syndrome or Neurotoxicity.
The Future of Lymphoma Management
The movement toward Polivy and Yescarta signifies the end of the “one size fits all” era of lymphoma treatment. We are entering a period of sequential precision, where the first line of defense is targeted, and the rescue therapy is cellular. As genomic sequencing becomes more integrated into routine care, the ability to predict which patient will respond to an ADC versus a CAR-T cell will further refine survival statistics.