As spring unfolds across Europe and North America, rising temperatures awaken blacklegged ticks carrying Borrelia burgdorferi, the bacterium responsible for Lyme disease. Early recognition of the characteristic erythema migrans rash—often appearing as a bull’s-eye lesion at the site of a tick bite within 3 to 30 days—remains the cornerstone of preventing progression to disseminated infection affecting joints, the nervous system, or the heart. Prompt antibiotic treatment, typically doxycycline for 10 to 21 days, achieves cure in over 90% of cases when initiated during this localized stage, according to current Infectious Diseases Society of America guidelines.
Understanding the Silent Spread: Why Early Detection Falters
Despite established protocols, Lyme disease frequently advances undetected because up to 30% of patients never recall a tick bite and the erythema migrans rash may lack the classic bull’s-eye appearance, presenting instead as a uniform red patch easily mistaken for a spider bite or allergic reaction. Dr. Élisabeth Baux, coordinator of the Eastern France Reference Center for Tick-Borne Diseases, emphasizes that “the absence of a remembered bite or atypical rash should not delay consideration of Lyme in endemic areas when patients present with unexplained flu-like symptoms, facial palsy, or knee swelling during peak tick season.” This diagnostic challenge contributes to the estimated 476,000 annual Lyme disease cases diagnosed and treated in the United States alone, a figure derived from insurance claims data analyzed by the Centers for Disease Control and Prevention (CDC).
In Plain English: The Clinical Takeaway
- Look for any expanding red rash at a tick bite site, even without the bull’s-eye pattern, especially if it appears within a month of outdoor exposure in wooded or grassy areas.
- Early flu-like symptoms—fatigue, fever, headache, or muscle aches—following a potential tick bite warrant medical evaluation, as antibiotics are most effective when started promptly.
- Prevention remains key: use EPA-registered insect repellents containing DEET or picaridin, perform full-body tick checks after outdoor activities, and shower within two hours to remove unattached ticks.
Geographic Expansion and Evolving Risk Maps
Lyme disease incidence is shifting northward and westward in response to climate change, with Ixodes scapularis and Ixodes pacificus ticks establishing populations in regions previously considered low-risk. In Canada, the Public Health Agency reports a tenfold increase in Lyme cases from 2009 to 2022, coinciding with the northward expansion of tick habitats into Ontario, Quebec, and the Maritime provinces. Similarly, in Europe, the European Centre for Disease Prevention and Control (ECDC) documents established transmission cycles now extending into southern Scandinavia and higher elevations in the Alps, driven by warmer temperatures prolonging tick activity seasons and increasing rodent reservoir host survival.
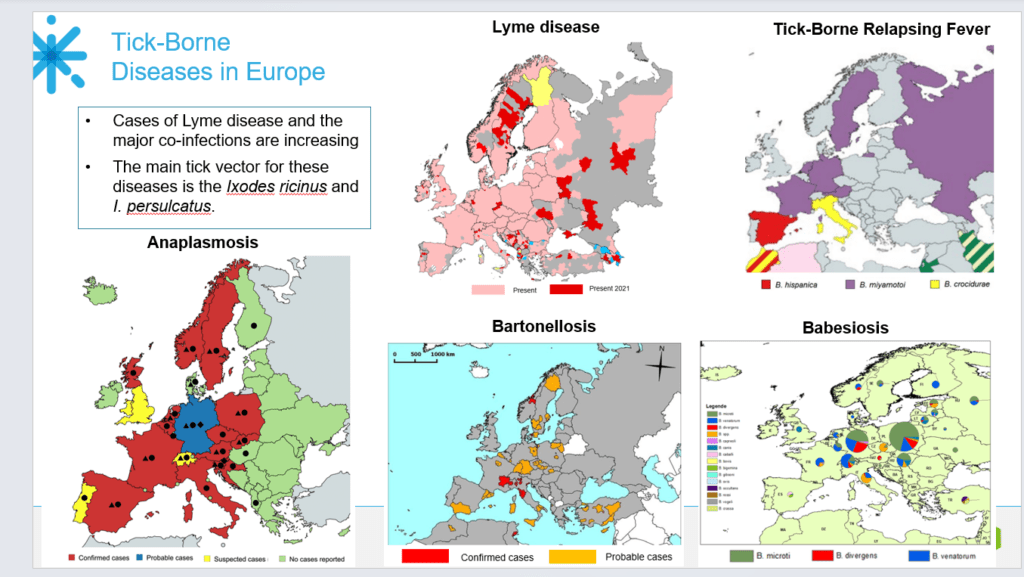

This geographic drift impacts healthcare access, as clinicians in newly endemic areas may lack familiarity with Lyme disease manifestations. In the United Kingdom, where the NHS reports approximately 1,000 laboratory-confirmed cases annually—likely an underestimate due to limited surveillance—public health agencies have issued guidance urging general practitioners in southern England to consider Lyme in patients with unexplained neurocognitive symptoms or migratory joint pain, even without travel history to classic endemic zones like the Scottish Highlands or New Forest.
Beyond Antibiotics: Research Frontiers and Therapeutic Gaps
While oral doxycycline, amoxicillin, or cefuroxime axetil remain first-line therapies for early Lyme disease, no vaccine is currently available for human use in North America or Europe following the withdrawal of LYMErix in 2002 due to low demand and unfounded safety concerns. Research into next-generation vaccines targets the outer surface protein A (OspA) of Borrelia burgdorferi, with a recombinant OspA vaccine candidate (VLA15) currently in Phase III clinical trials sponsored by Valneva and Pfizer. Preliminary data from Phase II trials, published in The Lancet Infectious Diseases, demonstrated robust antibody responses across multiple Borrelia serotypes with a favorable safety profile, supporting progression to efficacy testing in approximately 18,000 participants across endemic regions in the United States and Europe.
For patients experiencing persistent symptoms after recommended antibiotic courses—a condition termed Post-Treatment Lyme Disease Syndrome (PTLDS)—large longitudinal studies funded by the National Institutes of Health (NIH) have found no evidence of ongoing Borrelia infection and no benefit from prolonged antibiotic regimens. As Dr. John Aucott, Director of the Johns Hopkins Lyme Disease Clinical Research Center, states in a 2023 NIH-funded study:
“PTLDS represents a complex post-infectious phenomenon akin to other post-acute syndromes; our data show no microbiological evidence of persistent infection, and retreatment with antibiotics does not improve outcomes while carrying significant risks.”
Current management focuses on symptom-specific therapies, including graded exercise therapy for fatigue and cognitive behavioral therapy for pain-related distress, under the supervision of multidisciplinary teams familiar with the condition.
Contraindications & When to Consult a Doctor
Doxycycline is contraindicated in pregnant women, children under eight years due to risks of tooth discoloration and impaired bone growth, and individuals with known hypersensitivity to tetracyclines. Amoxicillin or cefuroxime axetil serve as preferred alternatives in these populations. Patients should seek immediate medical attention if they develop signs of disseminated Lyme disease, including severe headache with neck stiffness (suggesting meningitis), facial droop on one or both sides (facial palsy), palpitations or dizziness (indicating possible Lyme carditis), or large joint swelling—particularly the knee—persisting beyond a few days. Early intervention prevents progression to complications requiring intravenous antibiotics, such as ceftriaxone for neurologic or cardiac involvement.

The Path Forward: Integrating Surveillance and Public Education
Effective Lyme disease control hinges on integrating active tick surveillance with community education. The CDC’s TickNET collaboration network, comprising state health departments and academic centers, compiles real-time data on tick density and pathogen prevalence to inform targeted public alerts. In Europe, the ECDC promotes standardized monitoring through the VectorNet initiative, linking entomological findings with human case reporting across member states. Funding for these efforts derives primarily from government public health budgets, with supplemental support from entities like the NIH’s National Institute of Allergy and Infectious Diseases (NIAID) for vaccine research—a transparent model that mitigates industry bias concerns.
As outdoor recreation increases with warmer seasons, vigilance remains the best defense. Recognizing that early Lyme disease is highly treatable transforms anxiety into actionable awareness: a simple tick check after a hike or garden work, coupled with prompt attention to evolving rashes or flu-like symptoms, can avert months of preventable morbidity. The goal is not to instill fear of nature, but to empower individuals with the knowledge to enjoy it safely.
References
- Centers for Disease Control and Prevention. Lyme Disease Data and Statistics. Accessed April 2026. https://www.cdc.gov/lyme/stats/index.html
- Infectious Diseases Society of America. Clinical Practice Guidelines for Lyme Disease. Clin Infect Dis. 2020;71(1):e1-e45. https://doi.org/10.1093/cid/ciz1085
- Lantos PM, et al. Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA): 2020 Update on Diagnosis, Treatment, and Prevention of Lyme Disease. Clin Infect Dis. 2021;73(7):e1-e45. https://doi.org/10.1093/cid/ciab238
- Valneva and Pfizer. VLA15 Lyme Disease Vaccine Candidate Phase III Trial. ClinicalTrials.gov Identifier: NCT05477524. Accessed April 2026. https://clinicaltrials.gov/ct2/show/NCT05477524
- Aucott JN, et al. Post-Treatment Lyme Disease Syndrome Symptom Profile and Impact on Health-Related Quality of Life. Front Med (Lausanne). 2020;7:589. https://doi.org/10.3389/fmed.2020.00589

