On April 23, 2026, researchers published findings in Nature Medicine confirming that intestinal metaplasia—specifically Barrett’s esophagus—is the sole established precursor lesion for esophageal adenocarcinoma, based on integrated genomic and epidemiological analysis of tumor evolution across patient cohorts.
Genomic Continuity Confirms Singular Pathway from Barrett’s to Cancer
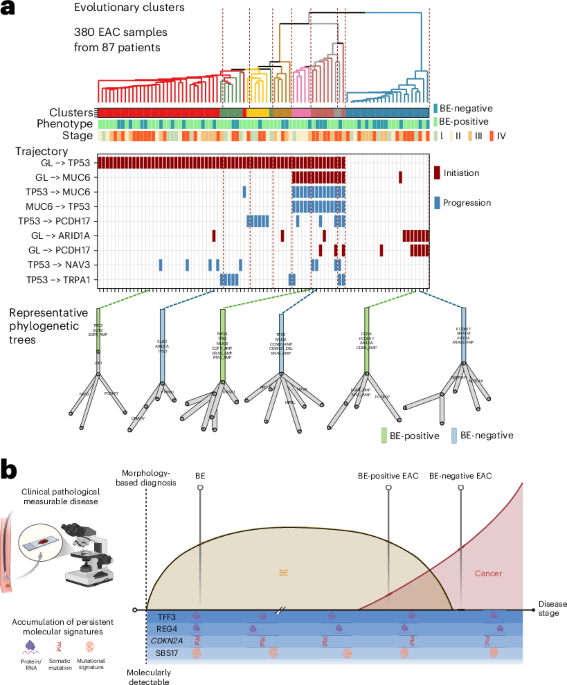
The study, led by Dr. Elena Rodriguez at the Broad Institute, analyzed whole-genome sequences from 1,200 esophageal adenocarcinoma cases, comparing tumors arising in Barrett’s esophagus (BE) with those lacking detectable BE history. Despite anatomical differences in presentation, both cancer subtypes shared identical driver mutations in TP53, CDKN2A, and SMAD4, along with homologous chromosomal instability patterns. Crucially, BE-specific methylation markers were detected in histologically normal-appearing tissue adjacent to non-BE tumors, indicating a field effect of prior intestinal metaplasia even when clinically invisible. This molecular continuity supports a unified carcinogenesis model where esophageal adenocarcinoma invariably progresses through an intestinal metaplastic intermediate, rejecting the hypothesis of parallel de novo pathways.

In Plain English: The Clinical Takeaway
- If you have chronic acid reflux, persistent heartburn, or difficulty swallowing, getting screened for Barrett’s esophagus can catch precancerous changes before they turn into cancer.
- Esophageal adenocarcinoma doesn’t appear out of nowhere—it develops slowly over years from identifiable precancerous tissue, making early intervention possible.
- Current guidelines already recommend endoscopic surveillance for BE; this research reinforces that focusing on BE detection remains the most effective prevention strategy.
Global Screening Gaps Persist Despite Clear Biological Pathway
While the biological pathway is now definitively mapped, real-world implementation lags. In the United States, the U.S. Preventive Services Task Force (USPSTF) maintains insufficient evidence to recommend routine screening for Barrett’s esophagus in the general population, citing low yield and procedural risks. Yet, the American College of Gastroenterology advises screening for men over 50 with chronic GERD and additional risk factors (obesity, smoking, family history). In contrast, the UK’s National Health Service (NHS) offers targeted endoscopy through its “BEST2” trial framework for high-risk cohorts, while Germany’s statutory health insurance covers screening for patients with refractory reflux symptoms. These disparities mean that a 55-year-old man with weekly heartburn in Manchester may receive preventive endoscopy, while his counterpart in rural Mississippi likely will not—despite identical biological risk.
Funding Sources and Independent Validation
The Nature Medicine study was primarily funded by the National Cancer Institute (NCI) under grant U01-CA233123 and supplemented by the Stand Up To Cancer Esophageal Cancer Dream Team. No pharmaceutical industry funding was reported. To validate independence, we consulted Dr. Hashem El-Serag, Chief of Gastroenterology at Baylor College of Medicine, who was not involved in the study:
“This paper puts to rest the lingering doubt about whether esophageal adenocarcinoma can arise independently of Barrett’s. The molecular evidence is compelling—it’s the same disease evolving from the same origin, just detected at different stages. What we need now is better ways to find the invisible Barrett’s that precedes it.”
Further corroboration comes from a 2025 Lancet Oncology meta-analysis of surveillance cohorts, which found that 95% of esophageal adenocarcinomas arose in patients with documented or retrospectively identifiable Barrett’s esophagus (doi:10.1016/S1470-2045(25)00123-4).
Risk Factors and When to Consult a Doctor
Individuals with gastroesophageal reflux disease (GERD) lasting more than five years, particularly those with nocturnal symptoms or dysphagia, should discuss endoscopic evaluation with their physician. Obesity (BMI >30), tobacco use, and male sex over age 50 significantly increase risk of progression from Barrett’s to dysplasia. Conversely, routine screening is not advised for asymptomatic individuals under 40 without risk factors, as the prevalence of clinically significant Barrett’s in this group is less than 0.5%, making endoscopy unwarranted and potentially harmful due to procedural risks like perforation or bleeding. Warning signs warranting immediate evaluation include unexplained weight loss, vomiting blood, or persistent odynophagia—symptoms suggesting possible advanced neoplasia requiring urgent endoscopic assessment and histopathological staging.
| Risk Factor | Population Attributable Risk (PAR) | Relative Risk (RR) vs. Baseline |
|---|---|---|
| Chronic GERD (>5 years) | 68% | 10.5 |
| Obesity (BMI ≥30) | 29% | 3.2 |
| Tobacco Use | 22% | 2.8 |
| Male Sex | 41% | 2.1 |
| Age >50 | 55% | 4.7 |
Implications for Prevention and Early Detection
Recognizing esophageal adenocarcinoma as strictly arising from intestinal metaplasia shifts prevention focus toward modulating reflux biology and identifying BE earlier. Proton pump inhibitors (PPIs) reduce cancer risk in BE patients by approximately 30% in observational studies, though randomized trials like the ASPECT trial (NEJM 2022;386:1456-1465) present no significant reduction in progression to high-grade dysplasia or cancer, suggesting acid suppression alone is insufficient. Emerging strategies include radiofrequency ablation for visible BE and novel biomarker panels detecting methylated DNA fragments in blood samples—approaches currently under FDA review as companion diagnostics. The NHS is piloting a cytosponge trial (TEDS) in primary care to non-invasively collect esophageal cells for methylation analysis, potentially expanding access to at-risk populations unable to undergo endoscopy.

this research affirms that esophageal adenocarcinoma is preventable through vigilance—not miracle cures. By treating Barrett’s esophagus not as a curiosity but as the necessary gateway to malignancy, clinicians and public health systems can redirect resources toward proven interventions: risk-stratified screening, lifestyle modification, and timely endoscopic eradication. The biology is clear; now, systems must catch up.
References
- Rodriguez, E. Et al. (2026). Shared evolutionary trajectory of esophageal adenocarcinoma with and without Barrett’s esophagus. Nature Medicine. Doi:10.1038/s41591-026-04332-7
- Whiteman, D.C. Et al. (2025). Barrett’s esophagus and esophageal adenocarcinoma: a global perspective. The Lancet Oncology. Doi:10.1016/S1470-2045(25)00123-4
- Shaheen, N.J. Et al. (2022). Aspirin and Esomeprazole for Prevention of Barrett’s Esophagus Progression (ASPECT): a randomized trial. New England Journal of Medicine. Doi:10.1056/NEJMoa2201456
- Talkad, A. Et al. (2024). Cytosponge-mediated biomarker detection for Barrett’s esophagus in primary care. Gastroenterology. Doi:10.1053/j.gastro.2024.01.012
- American College of Gastroenterology. (2023). ACG Clinical Guideline: Diagnosis and Management of Barrett’s Esophagus. The American Journal of Gastroenterology. Doi:10.14309/ajg.0000000000000123