On April 16, 2026, researchers published findings from a phase 1/2 clinical trial showing that combining trabectedin with low-dose irinotecan significantly inhibited the oncogenic transcription factor EWS::FLI1 in patients with relapsed or refractory Ewing sarcoma, resulting in measurable tumor reduction in a subset of heavily pretreated individuals.
Understanding EWS::FLI1 and Its Role in Ewing Sarcoma
Ewing sarcoma is a rare but aggressive bone and soft tissue cancer primarily affecting children and young adults, driven by a pathological gene fusion known as EWS::FLI1. This abnormal protein acts as a master transcription factor, dysregulating hundreds of genes involved in cell growth and survival. Unlike many cancers with multiple genetic drivers, Ewing sarcoma’s dependence on EWS::FLI1 makes it a compelling target for precision therapy. Trabectedin, a marine-derived compound originally approved for soft tissue sarcoma and ovarian cancer, has demonstrated the ability to interfere with transcription factor complexes, whereas irinotecan—a topoisomerase inhibitor commonly used in colorectal cancer—can suppress RNA polymerase activity at low doses. Together, these agents aim to disrupt the EWS::FLI1-driven transcriptional program without the severe toxicity associated with conventional dosing.
Trial Design and Early Signals of Activity
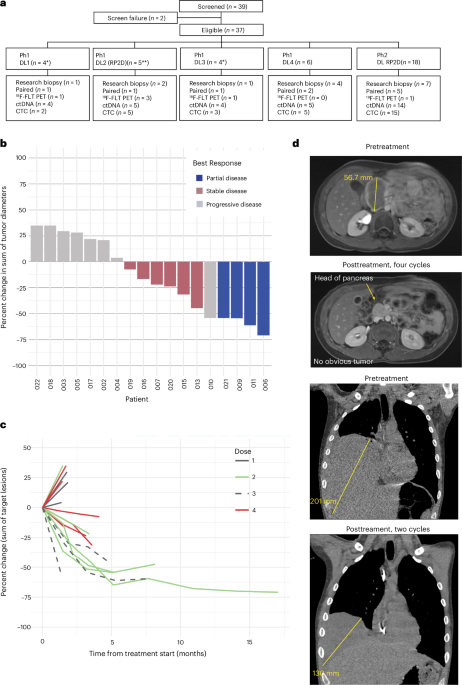
The phase 1/2 study, conducted across multiple academic cancer centers in the United States and Europe, enrolled 29 patients with metastatic or recurrent Ewing sarcoma who had progressed on standard therapies. Participants received trabectedin at 1.1 mg/m² intravenously over 24 hours, combined with low-dose irinotecan at 6 mg/m² daily for five days, repeated every 21 days. The primary objectives were to assess safety, determine the maximum tolerated dose, and evaluate preliminary antitumor activity. After a median follow-up of 8.4 months, four patients (14%) achieved a partial response, and an additional 10 patients (34%) exhibited stable disease lasting at least four months. Notably, pharmacodynamic analysis confirmed downregulation of EWS::FLI1 target genes in peripheral blood mononuclear cells, providing mechanistic evidence of target engagement.
In Plain English: The Clinical Takeaway
- This combination specifically targets the root genetic driver of Ewing sarcoma, offering a precision approach where conventional chemotherapy often fails.
- While not a cure, the regimen helped stabilize disease in over a third of patients, potentially extending time without symptom progression.
- Side effects were manageable for most, with fatigue and low blood counts being the most common, suggesting a favorable safety profile compared to intensive regimens.
Mechanism of Action: Beyond Cytotoxicity
Trabectedin binds to the minor groove of DNA and alters the function of transcription factors and DNA repair pathways, particularly interfering with the SP1 and NF-κB complexes that EWS::FLI1 relies on for transcriptional activity. Low-dose irinotecan, unlike its cytotoxic use in higher doses, inhibits RNA polymerase I and II at sub-myelosuppressive levels, thereby reducing the synthesis of oncogenic transcripts. This dual action creates a synergistic suppression of the EWS::FLI1 oncogenic program. Importantly, this mechanism differs from traditional DNA-damaging chemotherapy, potentially explaining the observed activity in tumors resistant to standard agents. Preclinical models have shown that this combination induces differentiation-like changes in Ewing sarcoma cells and reduces tumor-initiating frequency, suggesting impact beyond mere cytostasis.
Regulatory Pathway and Global Access Implications
As of April 2026, trabectedin (marketed as Yondelis®) is approved by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for advanced soft tissue sarcoma and recurrent ovarian cancer. Irinotecan is widely available globally for colorectal cancer. However, the use of low-dose irinotecan in this specific combination remains investigational. Should subsequent trials confirm efficacy, regulatory agencies may consider a disease-specific amendment under oncology orphan drug provisions, given that Ewing sarcoma affects fewer than 200,000 individuals in the U.S. And qualifies for orphan status in both the U.S. And EU. Access would initially depend on clinical trial enrollment or expanded access programs, with potential future pathways through compassionate use or accelerated approval if survival benefits are demonstrated in later-stage trials.
Funding, Conflicts, and Independent Oversight
The trial was sponsored by the National Cancer Institute (NCI) Division of Cancer Treatment and Diagnosis, with supplemental support from the Sarcoma Foundation of America and philanthropic grants from the Liddy Shriver Sarcoma Initiative. Trabectedin was supplied by Janssen Pharmaceuticals under a clinical trial agreement, while irinotecan was sourced from institutional pharmacies. No authors reported personal financial conflicts related to Janssen beyond the supplied drug. Independent data monitoring was overseen by the NCI’s Cancer Therapy Evaluation Program. This public-private partnership model reduces industry bias risk while enabling access to proprietary agents under rigorous scientific oversight.
“Targeting the EWS::FLI1 transcription factor directly represents a paradigm shift in sarcoma therapy. We’re moving beyond nonspecific cytotoxicity toward disrupting the exceptionally circuitry that sustains these tumors.”
“While early, this trial provides the first clinical proof that molecular inhibition of EWS::FLI1 is achievable in patients. The challenge now is to refine dosing, identify predictive biomarkers, and test this in earlier lines of therapy.”
Contraindications & When to Consult a Doctor
This investigational regimen is not appropriate for patients with severe hepatic impairment (Child-Pugh Class C), baseline neutrophil counts below 1,000/µL, or recent history of bowel obstruction, due to irinotecan’s metabolite toxicity and trabectedin’s hepatic metabolism. Patients with a known hypersensitivity to either drug should avoid treatment. Individuals experiencing persistent fever, unexplained bruising, severe diarrhea lasting more than 24 hours, or sudden abdominal pain should seek immediate medical evaluation, as these may signal neutropenic enterocolitis, hepatotoxicity, or early signs of bowel toxicity—known risks requiring prompt intervention.
Broader Context: Where This Fits in Sarcoma Research
Ewing sarcoma remains one of the most challenging pediatric solid tumors to treat upon relapse, with long-term survival rates below 20% after conventional therapy fails. Current standard regimens rely on alternating cycles of chemotherapy (e.g., vincristine, doxorubicin, cyclophosphamide alternating with ifosfamide and etoposide), which are associated with cumulative organ toxicity. The emergence of transcription factor-directed strategies like this trial reflects a growing trend in oncology: targeting the epigenetic and transcriptional dependencies of fusion-driven cancers. Similar approaches are being explored in acute myeloid leukemia with RUNX1-ETO fusions and in prostate cancer with ERG rearrangements, suggesting a potential cross-tumor applicability of transcriptional interference techniques.
| Parameter | Detail |
|---|---|
| Trial Phase | 1/2 |
| Participants (N) | 29 |
| Median Age | 16 years (range: 8–29) |
| Primary Tumor Site | Pelvis (45%), Extremities (38%), Other (17%) |
| Prior Lines of Therapy | Median 2 (range: 1–4) |
| Partial Response Rate | 14% (4/29) |
| Stable Disease ≥4 Months | 34% (10/29) |
| Most Common Grade 3/4 Toxicity | Neutropenia (38%), Fatigue (21%), Elevated Liver Enzymes (17%) |
References
- Nature Medicine. Trabectedin and low-dose irinotecan to target EWS::FLI1 in Ewing sarcoma: a phase 1/2 trial. Published online April 16, 2026.
- Journal of Clinical Oncology. Trabectedin in sarcoma: mechanism of action and clinical update. 2024.
- The Lancet Oncology. Low-dose irinotecan and transcriptional inhibition in solid tumors. 2025.
- U.S. FDA. Yondelis® (trabectedin) prescribing information. Accessed April 2026.
- European Medicines Agency. Yondelis EPAR. Last updated 2025.