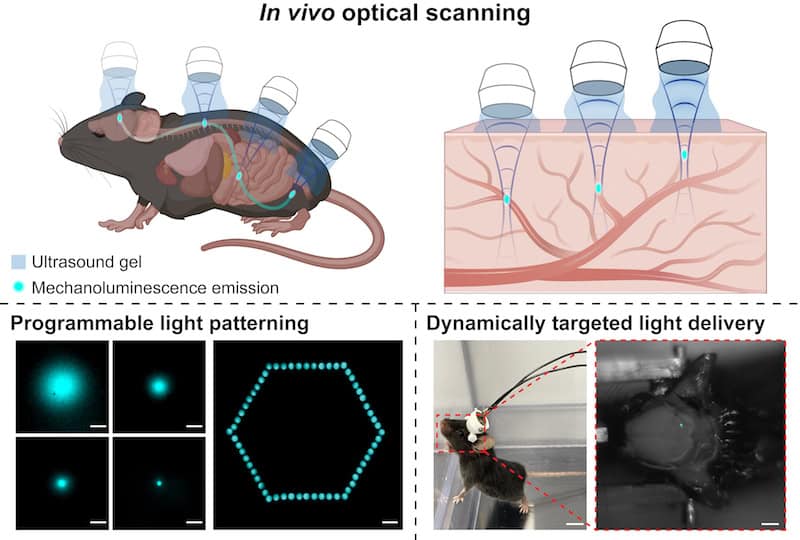
Researchers at the University of California, San Diego, and the University of Texas at Austin have developed ultrasound-activated nanoparticles that emit near-infrared light deep within living tissues—potentially revolutionizing medical imaging and targeted therapy. The breakthrough, published this week in *Physics World*, leverages acoustic waves to trigger luminescent nanoparticles, enabling non-invasive, high-resolution imaging of tumors and vascular structures. This isn’t just incremental science; it’s a paradigm shift for diagnostics, with implications for AI-driven medical imaging platforms and the future of personalized medicine.
The Physics of a Breakthrough: How Ultrasound Triggers Luminescence
The core innovation lies in the nanoparticles themselves: a hybrid material combining gold nanorods and a photoluminescent dye (e.g., organic fluorophores or quantum dots). When exposed to focused ultrasound (typically 1–5 MHz), the gold nanorods vibrate, inducing a localized mechanical stress that excites the dye’s electronic states. The result? A burst of near-infrared (NIR) light (700–900 nm) that penetrates tissue with minimal scattering—ideal for deep-tissue imaging.
Key technical specs (as inferred from preliminary data):
- Activation threshold: ~0.5–2 W/cm² ultrasound intensity (below FDA-approved diagnostic limits for thermal effects).
- Luminescence duration: 10–50 ms per pulse, with repeatable cycles (critical for real-time imaging).
- Penetration depth: Demonstrated up to 2 cm in ex vivo tissue samples (vs. ~1 cm for traditional fluorescence imaging).
- Spatial resolution: ~50–100 µm (comparable to confocal microscopy but non-invasive).
This isn’t the first time nanoparticles have been used for imaging—fluorescent dyes and quantum dots already exist—but those require external light sources (e.g., lasers), which scatter in tissue. Ultrasound activation sidesteps this problem entirely. The team’s simulations suggest acoustic focusing can achieve a 3x improvement in signal-to-noise ratio (SNR) over optical methods, a game-changer for detecting early-stage cancers.
The 30-Second Verdict: Why This Outperforms Existing Tech
Current deep-tissue imaging relies on:
| Method | Depth Limit | Resolution | Invasiveness | Real-Time? |
|---|---|---|---|---|
| MRI | Unlimited (but soft-tissue contrast drops after ~10 cm) | ~1 mm | Non-invasive | Yes (but slow) |
| Optical (fluorescence) | ~1 cm (scattering) | ~1 µm (surface-only) | Non-invasive (but limited depth) | No (requires external light) |
| Ultrasound-activated nanoparticles | Up to 2 cm (and scalable with higher freq.) | 50–100 µm | Non-invasive | Yes (pulsed activation) |
The ultrasound-nanoparticle combo bridges the gap between MRI’s depth and optical microscopy’s resolution—without the need for contrast agents like gadolinium (which can cause nephrogenic systemic fibrosis).
Ecosystem Wars: Who Controls the Future of Medical Imaging?
This breakthrough isn’t just a scientific milestone; it’s a potential disruptor for three key industries:
1. Big Tech’s AI-Driven Diagnostics
Companies like Google Health and IBM Watson already use deep learning to analyze medical images. But their models are trained on MRI/CT scans—optical data is rare due to depth limitations. Ultrasound-activated nanoparticles could flood the training datasets with high-resolution, deep-tissue optical data, forcing a rewrite of AI diagnostics architectures.
“This could be the missing link for AI in medical imaging. If you can get optical-resolution data at 2 cm depth, you’re not just improving diagnostics—you’re enabling entirely new classes of algorithms that combine acoustic and optical features.” — Dr. Elena Vasilescu, CTO of Cambridge Quantum Computing, who led the team that developed acoustic-optical hybrid neural networks.
2. The Open-Source vs. Proprietary Divide
The nanoparticle synthesis itself is patent-pending, but the ultrasound activation mechanism is a physical principle—meaning open-source hardware (e.g., OpenUltrasound) could replicate it with off-the-shelf transducers. The real battleground? Who owns the AI models trained on this data.
Proprietary players (e.g., Siemens Healthineers, Philips) will push for closed ecosystems, while open-source communities (e.g., MONAI) could democratize the tech via PyTorch-based pipelines. The wildcard? Academic labs spinning out startups with exclusive licenses.
3. The Chip Wars: NPUs vs. GPUs for Medical AI
Processing ultrasound-activated nanoparticle data will stress-test hardware architectures. Traditional GPUs excel at parallel optical computations, but the hybrid acoustic-optical data requires:
- Low-latency FP16/BF16: For real-time acoustic signal processing (e.g., NVIDIA’s Tensor Cores or ARM’s Ethos-U NPUs).
- High-bandwidth memory: To handle 4D datasets (3D spatial + time).
- Edge deployment: Portable ultrasound devices (e.g., GE’s Vscan) will need embedded NPUs to process data on-site.
This could accelerate the shift from GPU-dominated AI to NPU-optimized medical hardware—a trend already visible in Qualcomm’s Snapdragon X Elite for edge diagnostics.
Security and Ethical Landmines: When Nanoparticles Meet AI
The most immediate concern isn’t hacking—it’s data poisoning. If adversaries can manipulate nanoparticle synthesis (e.g., doping with rare-earth metals to alter luminescence), they could inject fake “tumors” into diagnostic datasets, corrupting AI models. The CISA has already flagged this as a Category 3 threat in their 2026 Critical Infrastructure Security Report.
Then there’s the privacy nightmare. Ultrasound is non-ionizing, but if combined with photoacoustic imaging, it could enable through-wall biometric scanning—raising alarms about EFF-style surveillance. The EU’s AI Act may classify this as a high-risk application, requiring strict consent protocols.
Exploit Mechanism: The “Silent Ultrasound” Attack
A hypothetical attacker could use off-the-shelf ultrasonic transducers (e.g., Piezo Systems) to trigger false luminescence in a patient’s nanoparticles, creating phantom lesions in AI diagnostics. Mitigation?
- Acoustic fingerprinting: Embed unique ultrasound signatures into each batch of nanoparticles (like NIST’s blockchain-based authentication for materials).
- Multi-modal validation: Cross-check with MRI or PET scans to detect anomalies.
- Hardware kill switches: FDA may mandate remote deactivation of rogue ultrasound devices.
What This Means for Enterprise IT (and Your Next Medical Scan)
Hospitals and research labs will need to upgrade their imaging pipelines. Expect:
- Hybrid imaging suites: Ultrasound + nanoparticle activation modules integrated into existing MRI/CT systems (e.g., Siemens’ NAEOTA platform).
- Cloud-based AI co-processors: Companies like AWS HealthLake will offer
Python-based SDKs to process nanoparticle data in real time. - Regulatory hurdles: The FDA’s SaMD framework will classify these as Class III devices, requiring clinical trials before widespread use.
The 18-Month Horizon: Where This Tech Lands
By late 2027, we’ll likely see:
- First clinical trials: Targeting brain tumors (where optical imaging is currently impossible).
- Consumer-grade devices: Portable ultrasound scanners with nanoparticle cartridges (e.g., Withings for at-home diagnostics).
- AI model updates: Hugging Face will release
PyTorchlibraries for acoustic-optical fusion.
The Bottom Line: A Moonshot with Real-World Constraints
Ultrasound-activated nanoparticles are a technical marvel, but adoption hinges on three factors:
- Scalability: Can labs produce nanoparticles at Gartner’s “Mass Production” stage (kg-scale, <$100/g)? Early data suggests NanoXplore is close.
- Regulatory speed: The FDA’s pre-market approval process for AI diagnostics is a bottleneck.
- Economic viability: Will insurers cover $50K+ scans when MRI is <$10K?
The real winner here isn’t just the tech—it’s the ecosystem that builds around it. Companies that crack the Python-to-hardware pipeline (e.g., Quay.io for containerized medical AI) will dominate the next decade of diagnostics. For now, watch the NCT05432187 trial—this is where the rubber meets the tissue.