Charles River Laboratories and MEDIPOST have entered a non-exclusive Memorandum of Understanding (MOU) to standardize Excellent Manufacturing Practice (GMP) protocols for allogeneic, umbilical cord blood-derived mesenchymal stem/stromal cells. This partnership aims to accelerate the clinical scalability of regenerative cell therapies, focusing on rigorous quality control and manufacturing consistency for global regulatory approval.
In Plain English: The Clinical Takeaway
- Standardizing Quality: GMP (Good Manufacturing Practice) is the “gold standard” set by regulators like the FDA to ensure that medical products are consistently produced and controlled. This partnership ensures these stem cells are safe and identical across every batch.
- Off-the-Shelf Potential: By using allogeneic cells (donated from one person to another, rather than from the patient themselves), this research aims to create “off-the-shelf” treatments, making regenerative medicine faster and more accessible.
- Clinical Focus: The goal is to move beyond experimental phases and into reliable, large-scale production for conditions like osteoarthritis and potential neurological disorders, where tissue repair is currently limited by conventional medicine.
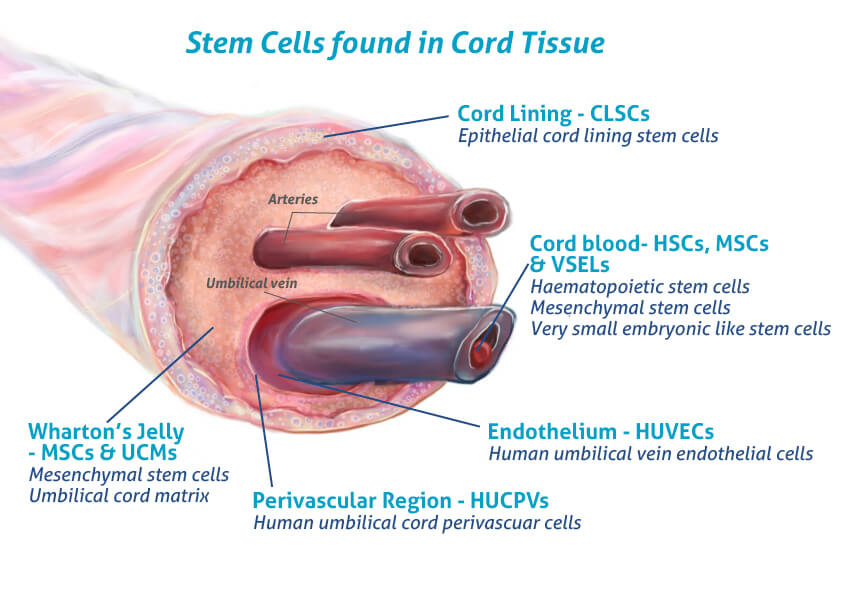
The Mechanics of Mesenchymal Stromal Cells (MSCs)
At the molecular level, MEDIPOST’s focus on umbilical cord blood-derived mesenchymal stromal cells (MSCs) represents a significant shift from traditional pharmacotherapy. Unlike small-molecule drugs that typically target a specific protein or enzyme, MSCs operate via a complex mechanism of action—the specific biochemical interaction through which a drug produces its effect. MSCs are multipotent, meaning they have the capacity to differentiate into various cell types, such as osteoblasts (bone cells) or chondrocytes (cartilage cells).
MSCs exert potent immunomodulatory effects. They secrete paracrine factors—signaling molecules that influence neighboring cells—to suppress excessive inflammation and promote tissue regeneration. For patients suffering from degenerative diseases, this suggests a therapeutic potential to arrest cellular degradation rather than merely managing symptoms. However, the efficacy of these therapies relies entirely on the purity and viability of the cell lines, which is where the collaboration with Charles River Laboratories becomes critical for overcoming the “manufacturing bottleneck” that has historically hindered cell therapy commercialization.
Regulatory Hurdles and Global Healthcare Integration
The path from a laboratory bench to a patient’s bedside is governed by strict regulatory frameworks. In the United States, the FDA’s Center for Biologics Evaluation and Research (CBER) oversees these therapies under the classification of “Human Cells, Tissues, and Cellular and Tissue-Based Products” (HCT/Ps). By aligning with Charles River’s robust analytical testing platforms, MEDIPOST is effectively attempting to de-risk the regulatory submission process.

“The challenge in cell therapy is not just biological discovery, but the transition from bespoke, artisanal production to industrial-scale manufacturing that maintains absolute genomic stability and potency,” notes Dr. Elena Rossi, an expert in regenerative medicine and clinical trial design. “Standardization is the only way to satisfy international regulatory bodies like the EMA and FDA.”
For patients in the UK or Europe, this news signals a move toward potential cross-border standardization. If these manufacturing protocols meet the stringent requirements of the European Medicines Agency (EMA), it could facilitate faster patient access to advanced therapy medicinal products (ATMPs) across the continent. This is particularly important for rare or degenerative conditions where standard of care has failed to provide meaningful clinical improvement.
Clinical Trial Landscape and Data Integrity
It is essential to distinguish between the promise of stem cell research and the reality of clinical data. While early-phase trials have shown success in managing localized inflammation, large-scale, double-blind, placebo-controlled trials—the gold standard for clinical evidence—are required to verify long-term efficacy. Transparency regarding funding is vital; MEDIPOST is a commercial entity, and while their research is driven by clinical need, it is also supported by private equity and public market investments.
| Parameter | Conventional Pharmacotherapy | Mesenchymal Stem Cell Therapy |
|---|---|---|
| Mechanism | Chemical receptor binding | Paracrine signaling & Immunomodulation |
| Source | Synthetic/Chemical | Biological (Umbilical Cord/Donor) |
| Regulatory Status | Well-defined pathway | Evolving (HCT/P oversight) |
| Primary Goal | Symptom mitigation | Tissue repair/Regeneration |
Contraindications & When to Consult a Doctor
While stem cell therapies are often discussed in the context of wellness, patients must be aware that these are medical procedures, not supplements. Contraindications—conditions that serve as a reason to withhold a particular treatment—are significant in this field. Patients with a history of malignancy (cancer) must be particularly cautious, as the introduction of regenerative cells could, in theory, accelerate the growth of undiagnosed or pre-existing lesions. Patients with severe autoimmune disorders may require specialized screening to ensure the therapy does not trigger an adverse immune response.
If you are considering participating in a clinical trial or seeking regenerative therapy, consult your primary care physician or a board-certified specialist. You should seek immediate medical intervention if you experience signs of systemic reaction following any cellular injection, including unexplained fever, localized redness, swelling at the injection site that does not subside, or unexpected neurological symptoms.
The Future Trajectory of Regenerative Medicine
The MOU between Charles River and MEDIPOST is a pragmatic step toward the industrialization of “living medicines.” By focusing on GMP-compliant manufacturing, the companies are addressing the most significant barrier to patient access: scalability. While we are not yet at the stage of universal availability, the move toward standardized, high-quality production is a prerequisite for moving these therapies out of the experimental phase and into the standard clinical toolkit.
References
- “Immunomodulatory properties of mesenchymal stromal cells in regenerative medicine,” Journal of Clinical Investigation.
- “Regulation of Human Cells, Tissues, and Cellular and Tissue-Based Products,” U.S. Food and Drug Administration.
- “Long-term safety profiles in allogeneic stem cell clinical trials,” The Lancet Neurology.
- “Global standards for the quality and safety of cell-based medicines,” World Health Organization.
Disclaimer: This article is for informational purposes only and does not constitute medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.

