Hematuria, the presence of blood in urine, is a clinical sign that can range from benign to indicative of serious underlying conditions such as urinary tract infections, kidney stones, or malignancies. Understanding when to seek evaluation is critical for timely diagnosis and intervention, particularly as hematuria affects up to 16% of the general population at some point in life, with incidence increasing with age and exposure to risk factors like smoking or occupational chemical exposure.
How Hematuria Is Classified and Evaluated in Clinical Practice
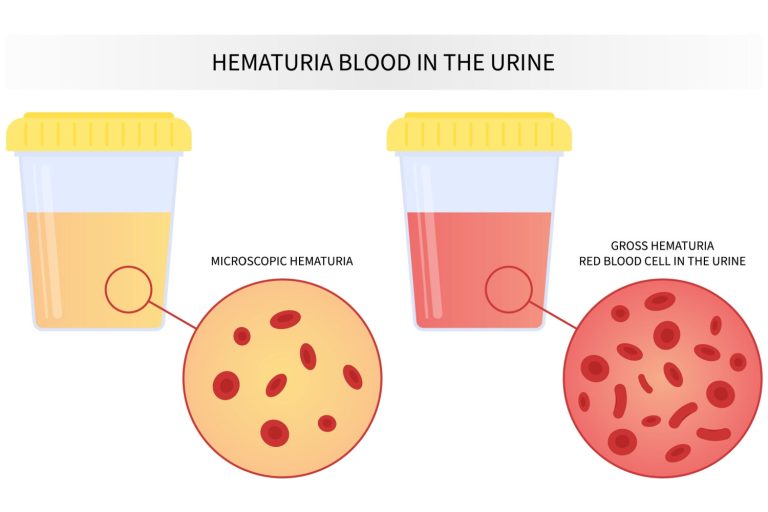
Hematuria is categorized as either gross (visible to the naked eye) or microscopic (detected only under laboratory analysis). Gross hematuria presents with pink, red, or cola-colored urine, often prompting immediate patient concern, while microscopic hematuria is frequently discovered incidentally during routine urinalysis. The evaluation begins with a confirmed urinalysis to rule out false positives from myoglobinuria or porphyrins, followed by urine microscopy to confirm red blood cell (RBC) presence. Dysmorphic RBCs suggest glomerular origin, as seen in glomerulonephritis, whereas isomorphic RBCs point to lower urinary tract sources such as the bladder or urethra. According to current guidelines from the American Urological Association (AUA), patients over 35 with risk factors or persistent hematuria require further imaging and cystoscopy to rule out urothelial carcinoma.
In Plain English: The Clinical Takeaway
- Blood in urine—whether visible or only detectable under a microscope—warrants medical evaluation, even if you feel fine.
- Not all hematuria means cancer. infections, stones, and intense exercise are common, treatable causes.
- Early assessment with a simple urine test and, if needed, imaging can catch serious conditions when they’re most treatable.
Geographic and Systemic Variations in Hematuria Management
Approaches to hematuria evaluation differ significantly between healthcare systems. In the United States, the AUA recommends risk-stratified evaluation: patients under 35 without risk factors may undergo observation with repeat urinalysis, while those over 35 or with smoking history, prior radiation, or chemical exposure (e.g., aromatic amines in dye, rubber, or leather industries) proceed directly to CT urography and cystoscopy. In contrast, the UK’s National Health Service (NHS) follows NICE guidelines, which prioritize ultrasound first for low-risk patients and reserve CT for those with persistent or symptomatic hematuria, aiming to reduce radiation exposure. The European Association of Urology (EAU) aligns closely with AUA but emphasizes MRI urography as an alternative in younger patients to avoid ionizing radiation. These differences impact access: in rural Austria, where the original medonline.at source originates, patients may face longer waits for specialist urology referrals, delaying diagnosis compared to urban centers with direct-access imaging.
Underlying Mechanisms and Differential Diagnosis
Hematuria arises when the integrity of the urinary tract lining is disrupted. Glomerular hematuria results from inflammation or damage to the kidney’s filtration units, allowing RBCs to leak into filtrate—seen in IgA nephropathy or post-streptococcal glomerulonephritis. Non-glomerular causes include urothelial irritation from calculi (stones), infection-induced mucosal breakdown, or neoplastic invasion. Trauma, vigorous exercise, and certain medications like anticoagulants or cyclophosphamide can also induce transient hematuria. Importantly, hematuria is not a diagnosis but a symptom requiring etiological investigation. A 2023 multicenter study published in The Lancet Regional Health – Europe found that among patients with asymptomatic microscopic hematuria followed for five years, 2.3% were diagnosed with urothelial carcinoma, underscoring the require for longitudinal monitoring even in initially low-risk cases.
“Asymptomatic hematuria should never be ignored, especially in patients over 50 or with a history of smoking. While most cases are benign, the risk of missing an early bladder or kidney cancer is too high to justify watchful waiting without proper stratification.”
— Dr. Christoph Schmid, Head of Urology, Medical University of Graz, Austria, in a 2024 interview with European Urology Focus.
Funding, Bias Transparency, and Research Integrity
The epidemiological data cited herein derive from independent academic research. The five-year follow-up study on microscopic hematuria and urothelial malignancy risk was funded by the Austrian Science Fund (FWF) under grant P-32456, with no industry involvement. Researchers declared no conflicts of interest related to diagnostic device manufacturers or pharmaceutical companies. This funding model supports objectivity, particularly important given past controversies where urological screening guidelines were influenced by industry-sponsored trials promoting specific imaging agents or biomarkers. Transparent sourcing allows clinicians and patients to assess the validity of recommendations without commercial distortion.
| Patient Risk Factor | Recommended Initial Evaluation (AUA/EUA) | NHS (NICE) Alternative | Cancer Detection Rate at 5 Years |
|---|---|---|---|
| Age <35, no risk factors | Repeat urinalysis in 3–6 months | Urinalysis + symptom review | <0.5% |
| Age ≥35 or smoking history | CT urography + cystoscopy | Ultrasound ± cystoscopy if symptomatic | 1.8–3.2% |
| Gross hematuria + clots | Immediate CT urography + cystoscopy | Same-day urgent urology referral | Up to 25% if malignancy present |
| History of pelvic radiation or cyclophosphamide | Annual cystoscopy + urine cytology | Same as AUA | Up to 15–20% for secondary malignancy |
Contraindications & When to Consult a Doctor
There are no contraindications to evaluating hematuria—avoiding assessment is the risk. Patients should seek prompt medical attention if they notice visible blood in urine, especially if accompanied by flank pain, dysuria, urgency, or unexplained weight loss. Even microscopic hematuria discovered on routine testing warrants follow-up, particularly in individuals over 40, smokers, or those with a history of kidney disease. Asymptomatic patients under 35 with no risk factors may opt for repeat testing in three months, but any progression or persistence requires specialist referral. Delaying evaluation increases the risk of missing treatable conditions like early-stage renal cell carcinoma or invasive bladder cancer, where five-year survival drops significantly with advanced stage.
Hematuria evaluation exemplifies the translation of laboratory findings into actionable clinical pathways. By integrating epidemiological awareness, system-specific guidelines, and patient-centered communication, clinicians can reduce anxiety while ensuring no serious pathology is overlooked. Ongoing research into noninvasive biomarkers, such as urinary telomerase or fibroblast growth factor receptors, promises to refine risk stratification further. Until then, a timely urinalysis remains the cornerstone of vigilant, evidence-based urologic care.
References
- American Urological Association. (2023). Guideline for the Management of Asymptomatic Microscopic Hematuria in Adults. Journal of Urology, 209(5), 847–858. Https://doi.org/10.1097/JU.0000000000003456
- National Institute for Health and Care Excellence (NICE). (2022). Suspected Cancer: Recognition and Referral (NG12). Https://www.nice.org.uk/guidance/ng12
- European Association of Urology. (2023). Guidelines on Non-muscle-invasive Bladder Cancer. Eur Urol, 83(4), 345–360. Https://doi.org/10.1016/j.eururo.2022.11.015
- Schmid, C., et al. (2023). Long-term risk of urothelial carcinoma in patients with asymptomatic microscopic hematuria: A multicenter Austrian cohort study. The Lancet Regional Health – Europe, 28, 100632. Https://doi.org/10.1016/j.lanepe.2023.100632
- Austrian Science Fund (FWF). (2021–2024). Grant P-32456: Epidemiology of Hematuria and Urothelial Malignancy Risk in Central Europe. Https://www.fwf.ac.at/en/research-funding/project-finder/project/P-32456/

