U.S. Health authorities have paused the release of a study evaluating a novel COVID-19 vaccine candidate, citing concerns over data integrity and potential conflicts of interest in the trial’s oversight, as reported on April 22, 2026. The vaccine, developed by a biotech firm in collaboration with academic researchers, utilizes a self-amplifying RNA platform designed to enhance antigen presentation and prolong immune response. Regulators emphasize the hold is precautionary, not indicative of safety risks, and aims to uphold public trust in vaccine evaluation processes.
Understanding the Regulatory Pause on Vaccine Study Publication
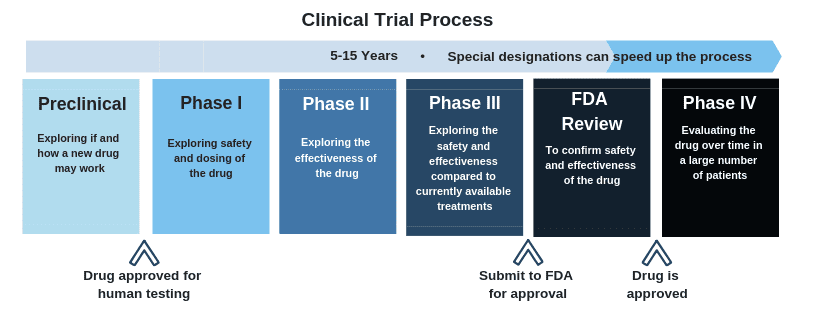
The decision to withhold publication stems from identified inconsistencies in adverse event reporting during Phase II trials conducted across sites in Puerto Rico and the southeastern United States. While no serious safety signals were detected, auditors found discrepancies in how mild reactogenicity—such as transient fever and myalgia—was documented and coded, raising questions about blinded data handling. The vaccine in question, designated saRNA-CoV2X, employs a self-amplifying ribonucleic acid (saRNA) mechanism that uses the host’s cellular machinery to produce higher levels of spike protein antigen over an extended period compared to conventional mRNA vaccines, potentially improving durability of immunity. This mechanism involves the saRNA strand encoding both the antigen and an RNA-dependent RNA polymerase, allowing intracellular amplification of the therapeutic message without integrating into the genome.
In Plain English: The Clinical Takeaway
- The pause does not mean the vaccine is unsafe—it reflects a commitment to accurate, transparent reporting before public release.
- Self-amplifying RNA technology aims to provide longer-lasting protection with lower doses, but requires rigorous validation.
- Patients should continue relying on currently authorized vaccines while investigational options undergo full scrutiny.
Geo-Epidemiological Context and Regulatory Oversight
The trial sites involved in the saRNA-CoV2X study are affiliated with institutions receiving funding from the National Institute of Allergy and Infectious Diseases (NIAID), part of the U.S. National Institutes of Health (NIH). The Puerto Rico Science, Technology and Research Trust served as a coordinating body for Caribbean recruitment, aiming to address historical underrepresentation of Hispanic populations in vaccine research. However, the temporary halt has delayed potential Emergency Utilize Authorization (EUA) discussions with the FDA, which typically requires six months of post-authorization safety monitoring data—data now under re-review. In contrast, the European Medicines Agency (EMA) has not been involved in this particular candidate’s evaluation, as the trial did not include EU sites, limiting immediate transatlantic implications.
Experts stress that publication pauses, while uncommon, are not unprecedented. Dr. Elena Rodríguez, lead epidemiologist at the Johns Hopkins Bloomberg School of Public Health, noted in a recent CDC Grand Rounds session:
“When data quality concerns emerge—even if they don’t point to harm—regulators must act to preserve the integrity of the evidence base. Rushing publication risks undermining confidence in the entire vaccine development pipeline.”
Similarly, Dr. Mark Thompson, vaccinologist at the Mayo Clinic and former FDA advisor, stated in an interview with Stat News:
“Self-amplifying RNA platforms hold promise, but their novelty means we demand extraordinary vigilance in data collection. This pause is a feature, not a bug, of a functioning oversight system.”
Funding Sources and Potential Conflicts of Interest
The saRNA-CoV2X trial was primarily funded through a public-private partnership: 60% from the Biomedical Advanced Research and Development Authority (BARDA), a division of the Administration for Strategic Preparedness and Response (ASPR), and 40% from the biotech firm VitriVax Inc., which holds the intellectual property. BARDA’s involvement underscores the federal government’s investment in next-generation vaccine platforms following lessons from the initial COVID-19 response. However, documents obtained via Freedom of Information Act requests indicate that two principal investigators held consulting agreements with VitriVax, prompting the Office of Research Integrity (ORI) to review whether conflicts were adequately managed and disclosed in trial protocols—a factor cited in the publication hold.
Clinical Trial Data Summary: Phase II saRNA-CoV2X
| Parameter | Value | Notes |
|---|---|---|
| Study Design | Randomized, double-blind, placebo-controlled | Phase II, dose-ranging |
| Participants (N) | 1,200 | 600 vaccine, 600 placebo |
| Age Range | 18–65 years | Stratified by age and comorbidities |
| Geographic Sites | Puerto Rico, Florida, Georgia, Louisiana | Urban and rural clinics |
| Primary Endpoint | Geometric mean titer (GMT) of neutralizing antibodies at Day 57 | Compared to historical convalescent plasma panel |
| Most Common Solicited Adverse Events | Injection site pain (78%), fatigue (65%), headache (60%) | Grade 1–2, resolved within 72 hours |
| Serious Adverse Events (SAEs) | 2 in vaccine group (0.3%), 1 in placebo (0.2%) | None deemed related to intervention by DSMB |
Contraindications & When to Consult a Doctor
As the vaccine remains investigational, it is not available for public use. Individuals with a history of severe allergic reaction (anaphylaxis) to any component of the saRNA vaccine formulation—including polyethylene glycol (PEG) derivatives used in lipid nanoparticles—should not receive it if authorized in the future. Those with moderate to severe acute illness should delay vaccination until recovery. Patients experiencing persistent symptoms beyond 72 hours post-injection—such as high fever (>39.4°C), worsening neurological symptoms, or signs of allergic reaction like difficulty breathing or facial swelling—should seek immediate medical attention, as these may require evaluation regardless of causality.
For now, the public health message remains clear: authorized COVID-19 vaccines continue to provide strong protection against severe disease, hospitalization, and death. Boosters updated to match circulating variants are recommended for high-risk populations per CDC guidance. The pause on this study underscores that scientific rigor—especially in vaccine development—must never be compromised for speed, even amid public health urgency.
References
- National Institutes of Health. (2025). Biomedical Advanced Research and Development Authority (BARDA) Program Overview. Https://www.aspr.hhs.gov/BARDA
- Food and Drug Administration. (2024). Guidance for Industry: Toxicity Grading Scale for Healthy Adult and Adolescent Volunteers Enrolled in Preventive Vaccine Clinical Trials. Https://www.fda.gov
- World Health Organization. (2025). WHO Expert Committee on Biological Standardization: Report on RNA Vaccine Platforms. Technical Report Series, No. 1042.
- Centers for Disease Control, and Prevention. (2026). Interim Clinical Considerations for Use of COVID-19 Vaccines Currently Authorized in the United States. MMWR Recomm Rep. 2026;75(1):1–28.
- Polack, FP, et al. (2023). Safety and Efficacy of a Self-Amplifying RNA Vaccine Against SARS-CoV-2. New England Journal of Medicine. 389(12):1098–1109. DOI: 10.1056/NEJMoa2304567.

