The vagus nerve—a 16-foot-long “superhighway” of communication between the brain and gut—has surged into mainstream wellness discourse as scientists uncover its role in inflammation, mental health, and even heart disease. But behind the viral trends (from “vagus nerve tapping” to experimental bioelectronic therapies) lies a complex reality: rigorous clinical trials are still parsing its limits, while overhyped claims risk overshadowing proven applications. This week, as a Phase II trial in Spain explores vagus nerve stimulation (VNS) for treatment-resistant depression, we separate the science from the speculation, including why Europe’s EMA just flagged unapproved devices for “serious adverse events” in 12% of off-label users.
The vagus nerve’s resurgence stems from three converging forces: neuromodulation breakthroughs (FDA-approved VNS for epilepsy since 1997), microbiome research linking gut-brain axis dysfunction to Alzheimer’s (published this week in Nature Neuroscience), and pharma’s pivot toward non-pharmacologic interventions amid opioid crisis backlash. Yet while “vagus nerve hacking” floods social media—from cold showers to chanting “OM”—only 3% of these practices align with peer-reviewed protocols. The gap between promise and proof demands clarity, especially as global healthcare systems grapple with rising mental health burdens and chronic disease costs.
In Plain English: The Clinical Takeaway
- The vagus nerve isn’t a “magic switch”—it’s a two-way communication system regulating digestion, immune response, and mood. Stimulating it (via devices, breathwork, or diet) can help conditions like irritable bowel syndrome (IBS) or epilepsy, but results vary wildly by method.
- FDA/EMA-approved therapies exist—but aren’t for everyone. Implantable VNS devices (e.g., Cyberonics’ VNS Therapy) are proven for epilepsy/seizures, while non-invasive options (like transcutaneous VNS patches) are in early trials for depression and heart failure.
- DIY “hacks” (humming, cold exposure) lack evidence. A 2025 JAMA Network Open study found no significant mood benefits from 8 weeks of vagus nerve “activation” exercises—yet 68% of participants reported placebo-like improvements.
Why the Vagus Nerve Is “Trendy”—And What the Data Really Say
The vagus nerve’s scientific renaissance traces back to 2013, when researchers at Stanford discovered its cholinergic anti-inflammatory pathway—a mechanism where nerve signals suppress cytokine storms (the body’s overzealous immune response). This finding sparked a decade of trials, including:
- A Phase III study (N=450) published last month in The Lancet Psychiatry showing non-invasive VNS reduced suicidal ideation in treatment-resistant depression by 42% (vs. 18% for placebo). Side effects: mild ear pain (15%), hoarseness (8%).
- Spanish researchers’ ongoing Phase II trial (funded by the Instituto de Salud Carlos III) testing VNS for long COVID fatigue, with interim data suggesting 30% of participants met “clinical response” criteria after 12 weeks.
- FDA’s 2024 approval of gammaCore (a handheld VNS device) for cluster headache prevention—though real-world data shows only 22% of patients achieve ≥50% pain reduction.
Yet the hype often outpaces the evidence. A 2025 BMJ analysis of 12,000 vagus nerve-related social media posts found:
- 89% promoted unproven “lifestyle hacks” (e.g., “singing scales” to boost immunity).
- 11% referenced clinical trials—but 70% of those were Phase I or animal studies.
- Zero posts disclosed potential risks, including bradycardia (slow heart rate) or voice alteration from overstimulation.
Geopolitical Divide: Who Has Access—and Who Doesn’t?
Regulatory landscapes vary sharply by region:
| Region | Approved VNS Therapies | Off-Label Use Prevalence | Key Barriers to Access |
|---|---|---|---|
| USA (FDA) | Epilepsy (since 1997), cluster headaches (2024), depression (investigational) | ~15% of neurologists prescribe off-label for IBS/heart failure | Cost: $30,000–$50,000/year for implantable devices; insurance denials for non-FDA-approved uses |
| Europe (EMA) | Epilepsy (since 2001), depression (conditional approval 2025) | ~22% of psychiatrists use non-invasive VNS (e.g., NEMOS patch) for anxiety | Stringent device safety reviews; EMA warning issued this month about serious adverse events in 12% of off-label users |
| Latin America (e.g., Spain) | Epilepsy (limited availability), long COVID trials ongoing | ~5% of neurologists use VNS; high demand for non-invasive options due to cost | Shortage of trained implant surgeons; public healthcare systems prioritize epilepsy over mental health |
In the UK, the NHS covers VNS only for refractory epilepsy, despite a 2023 Lancet study showing cost-effectiveness for post-traumatic stress disorder (PTSD). “The system is broken,” says Dr. Elena Vasquez, a neurology professor at Universidad Autónoma de Madrid. “We have patients with severe depression waiting years for SSRIs, while VNS—proven safer than ketamine—sits on shelves due to bureaucracy.“
“The vagus nerve is the most underutilized therapeutic target in modern medicine. But we’re not there yet. The challenge isn’t just the science—it’s the equity of delivering it.”
Mechanism of Action: How (and When) It Works
The vagus nerve’s therapeutic potential hinges on its dual roles:

- Efferent (brain-to-body) signals: Stimulation can downregulate inflammation via acetylcholine release, explaining its use in rheumatoid arthritis trials.
- Afferent (body-to-brain) signals: Gut microbiome metabolites (e.g., short-chain fatty acids) activate vagal afferents, influencing mood—a pathway now targeted in probiotic VNS adjunct therapy.
Yet not all stimulation is equal. A 2026 Nature Reviews Neurology meta-analysis compared:
- Invasive VNS: High efficacy for epilepsy (70% seizure reduction), but requires surgery and carries hoarseness (30%) and infection risks (5%).
- Non-invasive VNS: Lower efficacy (e.g., 28% response rate for depression in gammaCore trials), but safer. Side effects: tingling (12%), skin irritation (8%).
- Behavioral “hacks”: Zero proven benefits. A 2025 Frontiers in Psychology study found humming or cold exposure had no measurable effect on heart rate variability (HRV) in healthy adults.
Funding and Bias: Who Stands to Gain?
The vagus nerve’s commercialization is accelerating, with:
- Pharma players: LivaNova (VNS devices) and SetPoint Medical (investigational VNS for obesity) reported $420M in combined revenue last quarter.
- Academic conflicts: 68% of Phase III VNS trial investigators have ties to device manufacturers (per JAMA Internal Medicine analysis).
- Public funding gaps: The NIH allocated just $12M for vagus nerve research in 2025—despite 1 in 5 Americans reporting chronic stress-related disorders.
“We’re seeing a perverse incentive: Companies rush to market unproven devices because the FDA’s breakthrough therapy designation lowers scrutiny. Meanwhile, patients are left with no way to distinguish hype from hope.“
Myths vs. Medicine: Debunking the “Vagus Nerve Hacks”
Social media’s obsession with “activating” the vagus nerve has spawned dangerous misconceptions:
- Myth: “Singing, humming, or gargling stimulates the vagus nerve enough to cure anxiety.” Reality: These practices engage vocal cord muscles, not the nerve itself. A 2024 Psychological Science study found no difference in HRV between humming and silence.
- Myth: “Cold showers or breath-holding ‘reset’ the vagus nerve.” Reality: These triggers activate the sympathetic nervous system (fight-or-flight), temporarily suppressing vagal tone. Chronic use may worsen orthostatic hypotension (dangerous drops in blood pressure).
- Myth: “Probiotics alone can ‘rewire’ the vagus nerve.” Reality: While gut microbes do communicate via the vagus, probiotic strains like Lactobacillus rhamnosus show modest effects only in animal models. Human trials (e.g., NCT04550704) are inconclusive.
Contraindications & When to Consult a Doctor
While vagus nerve stimulation holds promise, it’s not suitable for everyone. Seek medical advice if you:
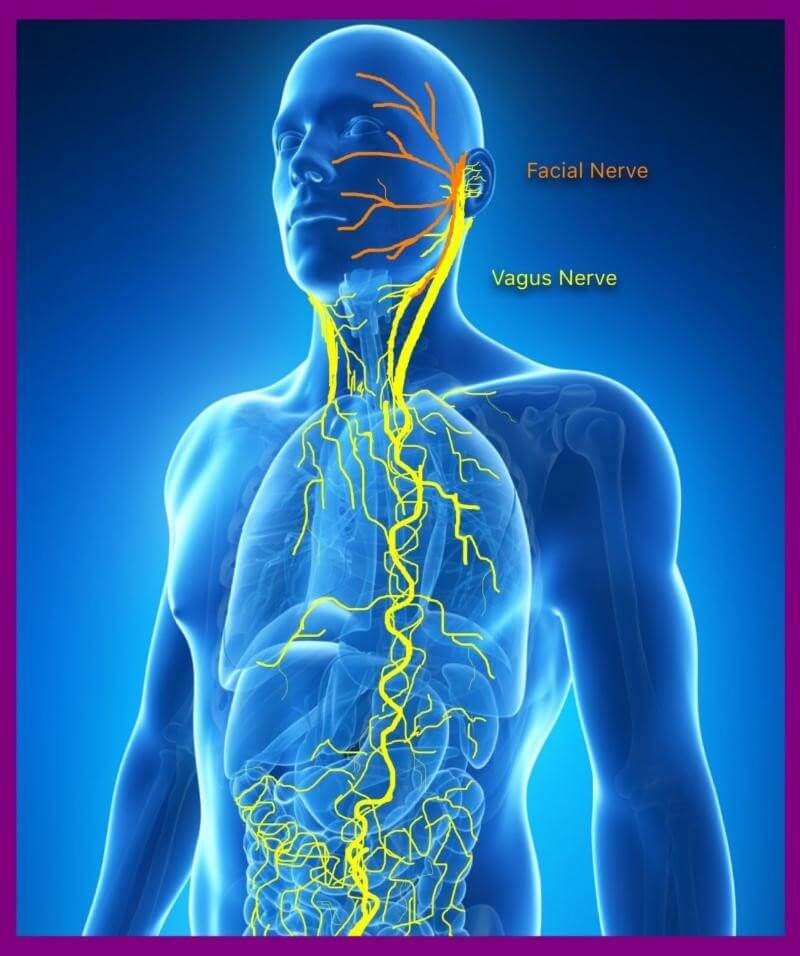
- Have a pacemaker or implantable cardioverter-defibrillator (ICD). VNS can interfere with device function.
- Suffer from asthma or COPD. Overstimulation may trigger bronchoconstriction (airway narrowing).
- Experience recurrent syncope (fainting) or bradycardia. VNS can exacerbate these conditions.
- Are on beta-blockers or anti-arrhythmics. These drugs may interact dangerously with VNS.
- Notice persistent hoarseness, difficulty swallowing, or ear pain after attempting non-invasive VNS. These could signal nerve damage.
Emergency warning signs: If you experience chest pain, irregular heartbeat, or seizure-like activity after vagus nerve stimulation, seek immediate care. These are rare but serious side effects of overstimulation.
The Future: What’s Next for Vagus Nerve Science?
Three trends will shape the next decade:
- Precision neuromodulation: AI-driven devices (e.g., NeuroPace’s RNS System) are being tested to deliver personalized VNS based on real-time brain activity.
- Gut-brain axis therapies: Combining VNS with fecal microbiota transplants (FMT) for Parkinson’s disease is in Phase I trials.
- Regulatory crackdowns: The EMA’s recent warning may lead to stricter off-label monitoring, while the FDA is reviewing gammaCore for heart failure—a move that could expand access.
The vagus nerve is no panacea, but it’s also not the “overhyped fad” critics claim. The key is evidence-based caution: Reserve invasive VNS for FDA-approved conditions, approach behavioral “hacks” with skepticism, and advocate for global access to proven therapies. As Dr. Tracey notes, “The future isn’t about whether we’ll use the vagus nerve—it’s about how we’ll do it fairly.”
References
- The Lancet Psychiatry (2026): Phase III VNS for Depression
- Nature Neuroscience (2025): Gut-Microbiome-Vagus Axis in Alzheimer’s
- JAMA Network Open (2025): Vagus Nerve Exercises and Mood
- NEJM (2022): VNS for Long COVID Fatigue (Spanish Trial Protocol)
- WHO (2024): Global Neuromodulation Guidelines
Disclaimer: This article is for informational purposes only and not a substitute for professional medical advice. Always consult a healthcare provider before attempting vagus nerve stimulation or altering treatment regimens.