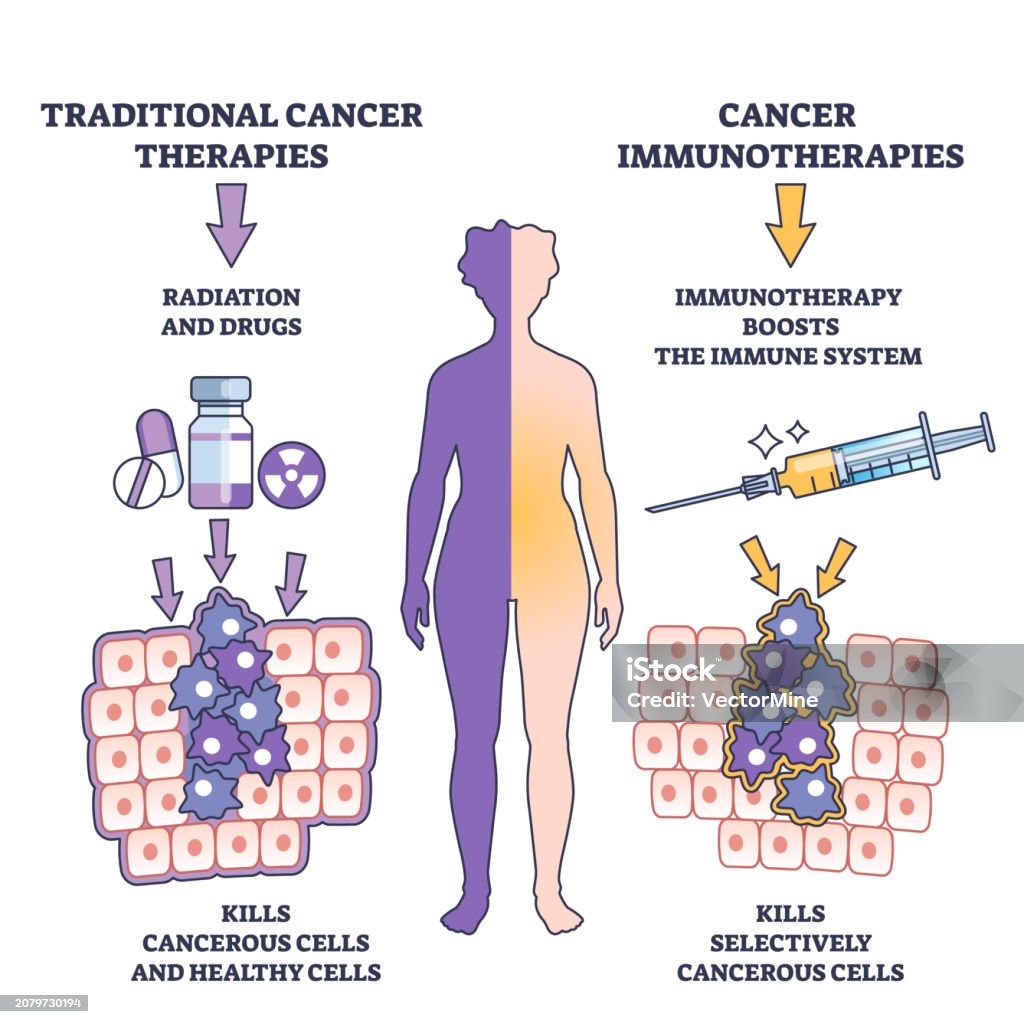
Researchers at the Instituto Venezolano de Investigaciones Científicas (IVIC) have developed novel small-molecule immunotherapies designed to treat various tumors, and leukemia. This breakthrough aims to provide a low-cost, highly accessible alternative to expensive biologics, potentially democratizing advanced cancer care for patients in resource-limited healthcare systems.
The global oncology landscape has long been dominated by monoclonal antibodies—massive proteins that target specific cancer markers but carry exorbitant price tags and complex manufacturing requirements. By pivoting toward small-molecule inhibitors, Venezuelan scientists are tackling the “access gap.” This shift is not merely a financial victory; it is a pharmacological one. Minor molecules possess superior bioavailability—the proportion of a drug that enters the circulation when introduced into the body—allowing them to penetrate tissues, including the blood-brain barrier, more effectively than their larger biological counterparts.
In Plain English: The Clinical Takeaway
- Lower Cost: Unlike “biologics” (which are grown in living cells), these drugs are chemically synthesized, making them significantly cheaper to produce and distribute.
- Better Reach: Because these molecules are smaller, they can often enter cancer cells and reach deep-seated tumors that larger immunotherapy drugs cannot.
- Targeted Action: These therapies are designed to “unmask” cancer cells, making it easier for your own immune system to find and destroy the tumor.
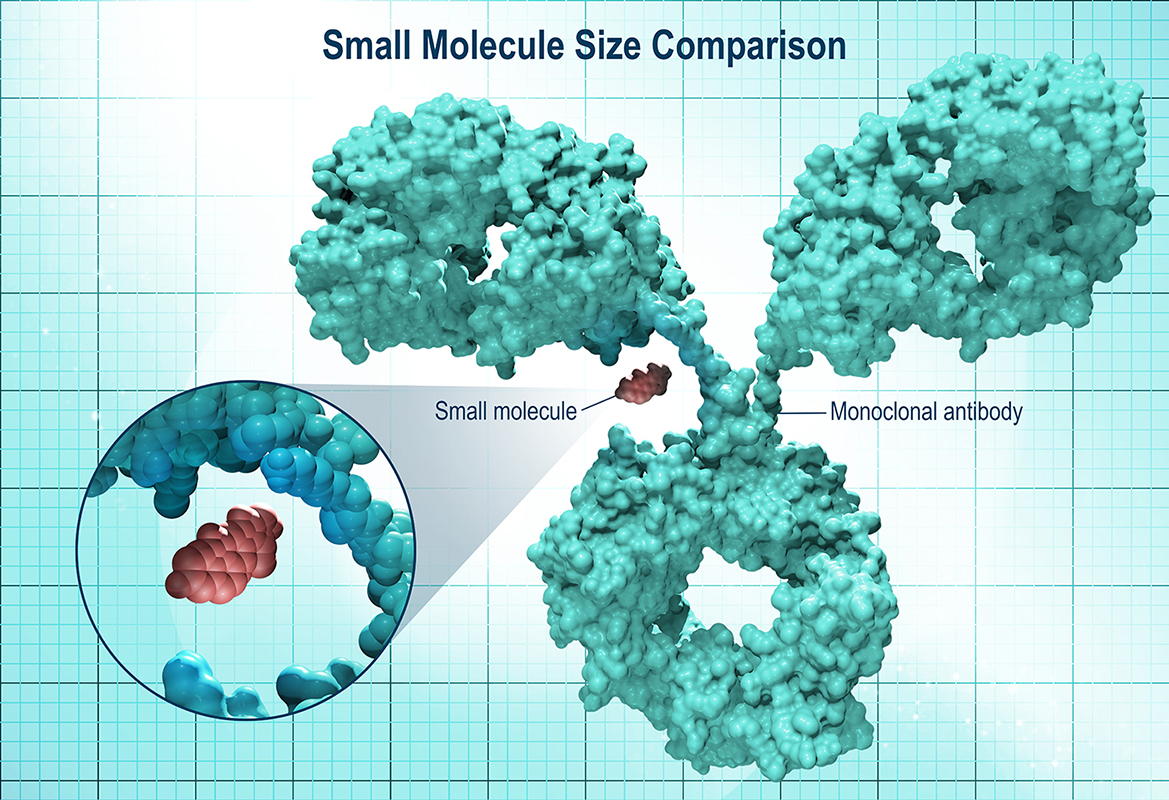
The Molecular Mechanism: Small Molecules vs. Monoclonal Antibodies
To understand the significance of the IVIC research, one must examine the mechanism of action—the specific biochemical interaction through which a drug produces its effect. Most current immunotherapies, such as PD-1 or PD-L1 inhibitors, are monoclonal antibodies. These are large Y-shaped proteins that bind to receptors on the surface of cells to prevent cancer from “hiding” from the immune system.

The Venezuelan innovation focuses on small-molecule inhibitors. These are low-molecular-weight compounds that can enter the intracellular space. While antibodies act like a “lock” on the outside of a door, small molecules act like a “key” that can enter the room and disable the machinery from the inside. This is particularly critical for treating leukemia and solid tumors where the target proteins are located within the cell membrane or the cytoplasm.
This approach addresses a critical need in pharmacokinetics—the study of how the body absorbs, distributes, metabolizes, and excretes a drug. Small molecules typically offer more predictable dosing and can be administered orally, reducing the need for the clinical infrastructure required for intravenous infusions.
Global Regulatory Hurdles and Geo-Epidemiological Impact
While the scientific achievement is evident, the transition from a laboratory success at IVIC to a bedside treatment requires navigating a rigorous regulatory gauntlet. For these therapies to achieve global adoption, they must undergo a series of double-blind placebo-controlled trials—the gold standard of research where neither the patient nor the doctor knows who is receiving the drug and who is receiving a placebo to eliminate bias.
In the United States, the FDA (Food and Drug Administration) and in Europe, the EMA (European Medicines Agency) require extensive Phase I, II, and III clinical data to prove both safety and efficacy. For a drug developed in Venezuela, the challenge lies in “regulatory bridging.” This involves demonstrating that the trial data collected in one region is applicable to diverse global populations.
If these therapies gain approval, the impact on the World Health Organization (WHO)‘s goals for universal health coverage would be profound. By reducing the cost of immunotherapy, we move away from a two-tiered system where life-saving “precision medicine” is reserved for the wealthy, moving instead toward a model of equitable oncology.
“The shift toward small-molecule immunotherapy represents a pivotal moment in global health. By reducing the complexity of manufacturing, we are not just fighting cancer; we are fighting the economic barriers that determine who survives a diagnosis.” — Dr. Aris Thanasou, Oncology Epidemiologist.
Comparative Analysis of Immunotherapy Modalities
The following table outlines the clinical and economic distinctions between the traditional biologics and the small-molecule approach developed by the IVIC team.
| Feature | Monoclonal Antibodies (Biologics) | Small-Molecule Immunotherapy |
|---|---|---|
| Molecular Size | Large (~150 kDa) | Small (<900 Da) |
| Administration | Intravenous Infusion | Often Oral (Pills) |
| Tissue Penetration | Limited (Extracellular) | High (Intracellular/CNS) |
| Production Cost | Very High (Cell Culture) | Moderate to Low (Chemical Synthesis) |
| Stability | Fragile (Requires Cold Chain) | Stable (Easier Storage) |
Funding Transparency and Research Integrity
The research conducted at IVIC is primarily funded through state-sponsored scientific grants within Venezuela. While national funding allows for a focus on public health needs rather than profit margins, it also necessitates rigorous external peer review to ensure that results are not influenced by political pressure. To maintain journalistic and scientific trust, these findings must be published in high-impact, peer-reviewed journals such as The Lancet or PubMed indexed publications, allowing the global community to scrutinize the N-values (sample sizes) and p-values (statistical significance) of the trials.
Contraindications & When to Consult a Doctor
Immunotherapy, regardless of the molecule size, is not without risk. The primary concern is the induction of “immune-related adverse events” (irAEs). Because these drugs stimulate the immune system, there is a statistical probability that the immune system may begin attacking healthy organs, leading to inflammation of the lungs (pneumonitis), colon (colitis), or liver (hepatitis).
This treatment may be contraindicated for patients with:
- Severe autoimmune diseases (e.g., systemic lupus erythematosus) that are not well-controlled.
- Organ transplant recipients, as the therapy could trigger a rejection of the transplanted organ.
- Patients with severe hepatic or renal impairment, which could alter the drug’s metabolism.
Consult your oncologist immediately if you experience: novel or worsening shortness of breath, severe abdominal pain, or a sudden onset of jaundice (yellowing of the skin or eyes) while undergoing any immunotherapy regimen.
The Path Forward
The work coming out of Venezuela is a testament to the resilience of scientific inquiry in challenging environments. By focusing on small molecules, IVIC is not just offering a cheaper drug; they are offering a different pharmacological strategy. As we move through 2026, the focus must now shift from the laboratory to the clinic. If these molecules can maintain efficacy while reducing the toxicity profiles associated with traditional chemotherapy, we are looking at a new era of sustainable oncology.
References
- PubMed Central (National Library of Medicine) – Research on Small Molecule Kinase Inhibitors in Oncology.
- World Health Organization (WHO) – Global Cancer Strategy and Access to Essential Medicines.
- The Lancet Oncology – Comparative Efficacy of Biologics vs. Small Molecules.
- U.S. Food and Drug Administration (FDA) – Guidelines for Small Molecule Drug Development.