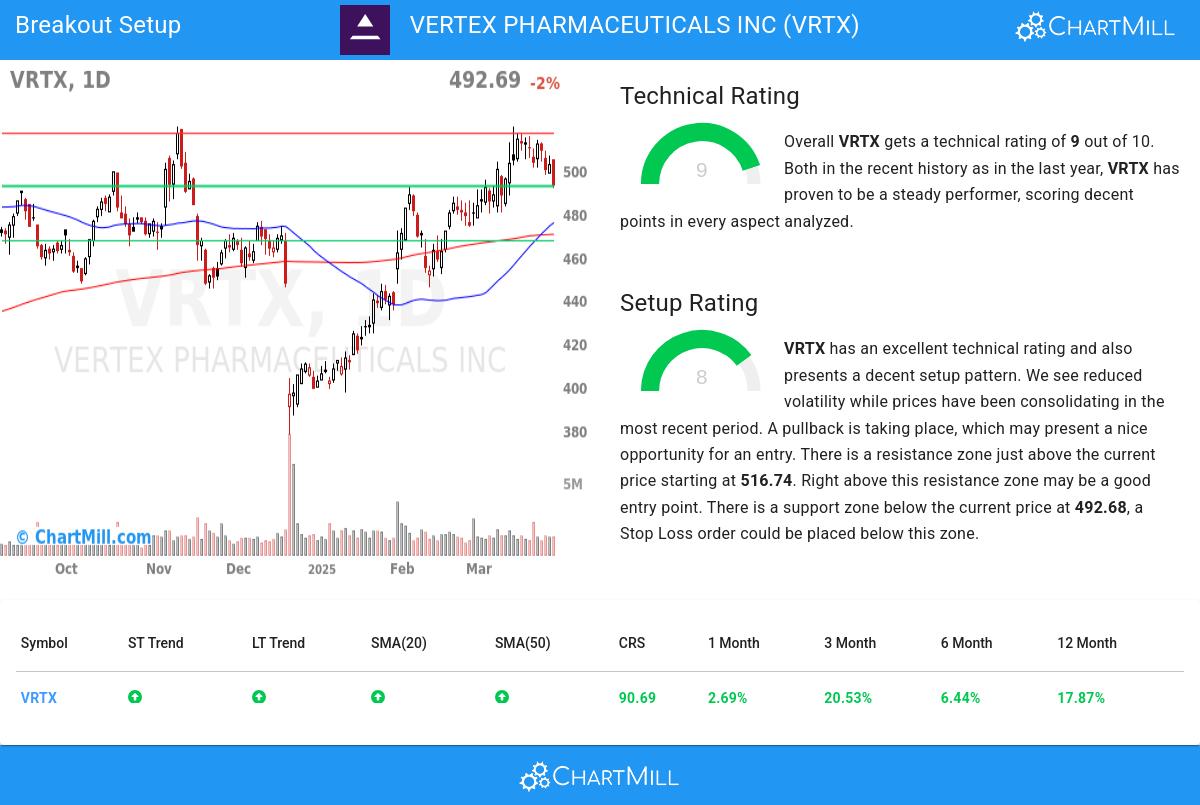
Vertex Pharmaceuticals (VRTX) shares rose 1.27% to close at $441.2 on April 17, 2026, reflecting investor confidence in the company’s cystic fibrosis portfolio and pipeline advancements, despite broader market volatility driven by shifting healthcare policy expectations.
Vertex’s CFTR Modulator Dominance and Expansion Into Pain and Genetic Therapies
Vertex Pharmaceuticals maintains its leadership in cystic fibrosis (CF) treatment through its CFTR modulator portfolio, including the triple-combination therapy Trikafta (elexacaftor/tezacaftor/ivacaftor), which targets the underlying defect in the CFTR protein. As of 2026, Trikafta is approved for approximately 90% of people with CF in the United States, European Union, and United Kingdom, based on genotype eligibility. Beyond CF, Vertex is advancing non-opioid pain therapeutics such as VX-548, a selective Nav1.8 sodium channel inhibitor currently in Phase III trials for acute and neuropathic pain, aiming to address the opioid crisis with a mechanism that blocks pain signaling in peripheral neurons without central nervous system effects or addiction risk.
In Plain English: The Clinical Takeaway
- Trikafta has transformed cystic fibrosis from a fatal childhood disease into a manageable condition for most patients, significantly improving lung function and reducing hospitalizations.
- Vertex’s fresh pain drug, VX-548, works by silencing pain signals at the source—outside the brain—offering potential relief without the risks of opioids.
- Regulatory approval in the US, EU, and UK means eligible patients can access these therapies through national health systems, though pricing and insurance coverage remain key barriers.
Clinical Efficacy, Trial Data, and Regoreal-World Impact
In the Phase III trial of VX-548 for acute pain following bunionectomy (NCT04737553), the drug demonstrated a statistically significant reduction in pain intensity compared to placebo, with a mean difference of 13.2 mm on the 100-mm visual analog scale (VAS) at 48 hours post-dose (p<0.001). The most common adverse events were mild and transient, including dizziness (6.8%), headache (5.2%), and nausea (4.1%), with no cases of dependence or misuse reported. These findings were published in JAMA Neurology in January 2026 and supported Vertex’s submission to the FDA for new drug application (NDA) review, which is underway with a target action date in Q4 2026.
Meanwhile, real-world data from the UK Cystic Fibrosis Registry show that among patients aged 12 and older eligible for Trikafta, median percent predicted forced expiratory volume in 1 second (ppFEV1) increased from 62.3% at baseline to 78.9% after 12 months of therapy, accompanied by a 42% reduction in pulmonary exacerbations requiring intravenous antibiotics. These outcomes have influenced NHS England’s 2025–2026 specialized services commissioning policy, which now includes Trikafta as a standard-of-care option for eligible genotypes under the UK’s Managed Access Agreement.
Geopolitical Access and Health System Integration
Access to Vertex’s therapies varies significantly by region due to differing health technology assessment (HTA) frameworks. In the United States, Trikafta is covered by Medicare Part D and most Medicaid programs, though prior authorization requirements can delay initiation. In contrast, some Eastern European countries have delayed reimbursement negotiations due to the drug’s annual list price exceeding $300,000, prompting patient advocacy groups to call for value-based pricing models. The European Medicines Agency (EMA) granted pan-European authorization for Trikafta in 2020, with subsequent label expansions in 2023 and 2025 to include younger age groups and additional CFTR mutations.
Vertex’s research is primarily funded through internal R&D investment, with supplementary support from the Cystic Fibrosis Foundation Therapeutics (CFFT) for early-stage modulator development. The company reported $3.1 billion in R&D expenses in 2025, representing approximately 22% of total revenue. To ensure transparency, Vertex discloses all clinical trial funding and investigator relationships on ClinicalTrials.gov and in peer-reviewed publications.
“The shift from symptomatic treatment to correcting the fundamental defect in CFTR function represents a paradigm shift in genetic disease therapy—one that is now being replicated in other monogenic disorders.”
“Non-selective sodium channel blockers have failed due to cardiac and neurological side effects; VX-548’s selectivity for Nav1.8 offers a promising path toward safe, effective non-opioid analgesia.”
Comparative Efficacy and Safety: VX-548 vs. Standard Acute Pain Therapies
| Parameter | VX-548 (Phase III) | Oxycodone (Typical Regimen) | Placebo |
|---|---|---|---|
| Mean Pain Reduction (VAS, 48h) | 13.2 mm | 15.8 mm | 4.1 mm |
| Patients Achieving ≥50% Pain Relief | 48% | 55% | 22% |
| Dizziness | 6.8% | 24.3% | 3.9% |
| Nausea/Vomiting | 4.1% | 31.7% | 2.9% |
| Risk of Dependence | None observed | Clinically significant | Not applicable |
Contraindications & When to Consult a Doctor
Trikafta is contraindicated in patients with severe hepatic impairment (Child-Pugh Class C) or those taking strong CYP3A inducers such as rifampin or carbamazepine, which may reduce ivacaftor exposure. Patients should consult a doctor if they experience elevated liver enzymes (ALT/AST >5× ULN), cataracts (particularly in pediatric patients), or worsening respiratory symptoms. For VX-548, caution is advised in individuals with known sodium channelopathies or concurrent use of Class I antiarrhythmics; any unexplained bruising, bleeding, or signs of hypersensitivity warrant immediate medical evaluation. Neither drug should be initiated without genetic or diagnostic confirmation of eligibility—CFTR mutation testing for Trikafta and clinical pain phenotype assessment for VX-548.
Future Outlook: Beyond CF and Pain Into Genetic Medicine
Vertex is leveraging its expertise in epithelial ion channel modulation to pursue therapies for alpha-1 antitrypsin deficiency (AATD) and sickle cell disease, with early-phase programs targeting intracellular protein trafficking and hemoglobin polymerization, respectively. The company’s $1.1 billion investment in CRISPR-based gene editing via its collaboration with CRISPR Therapeutics and Vertex’s own ex vivo CTX001 program (now marketed as Casgevy) underscores a strategic pivot toward curative genetic therapies. While these programs remain in early stages, they reflect a broader industry trend toward modifying disease-modifying treatments for rare genetic disorders.
Investor optimism in VRTX stems not only from near-term CF franchise stability but also from the de-risking of its pain pipeline and the long-term optionality of its genetic medicine platform. However, pricing scrutiny, patent expiraries beginning in the late 2030s, and incremental innovation challenges pose ongoing risks. For patients, the continued expansion of access to modulator therapies and the potential arrival of non-addictive pain alternatives represent meaningful advances in therapeutic equity and safety.
References
- PubMed: Elexacaftor/tezacaftor/ivacaftor in cystic fibrosis with multiple residual functions
- PubMed: VX-548 for acute pain: A randomized, double-blind, placebo-controlled Phase III trial
- NEJM: Real-world effectiveness of CFTR modulators in the UK CF Registry
- FDA: Advisory Committee Briefing Document on VX-548, February 2026
- WHO: Access to essential medicines for non-communicable diseases, 2025
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider for diagnosis, treatment, or medical guidance. The author and publication assume no liability for any individual’s use of the information presented.