Genetic disorders are rising globally, with India reporting a 22% increase in congenital conditions over the past decade, yet newborn screening programs remain underfunded. A breakthrough in preimplantation genetic testing (PGT) during IVF—now performed on Day 5 embryos—could revolutionize early detection, but access hinges on regulatory approval and public health infrastructure. Fertility specialist Dr. Rubina Pandit confirms the timing (Day 5 post-fertilization) aligns with blastocyst-stage embryo viability, but critical gaps remain in cost, equity, and long-term data.
Why This Matters: The Global Crisis of Undetected Genetic Disorders
Genetic disorders account for 30% of neonatal deaths worldwide, yet 98% of newborns in low-resource settings lack screening [WHO, 2025]. In India, conditions like Duchenne muscular dystrophy (X-linked recessive) and cystic fibrosis (autosomal recessive) often go undiagnosed until irreversible symptoms emerge. Preimplantation genetic testing (PGT), now optimized for Day 5 blastocysts, offers a window to prevent these disorders—but only if integrated into IVF protocols. The challenge? Cost ($10,000–$20,000 per cycle) and regulatory fragmentation across regions like the EU (EMA) and US (FDA).
In Plain English: The Clinical Takeaway
- What it is: PGT tests embryos created via IVF for genetic disorders before implantation, using a tiny biopsy of cells from a 5-day-old blastocyst (a hollow ball of cells).
- Why it matters: Catches disorders like Down syndrome, sickle cell anemia, or Tay-Sachs before pregnancy, reducing heartbreak and medical costs later.
- The catch: Only works for couples using IVF (not natural conception) and isn’t covered by most insurance, even in high-income countries.
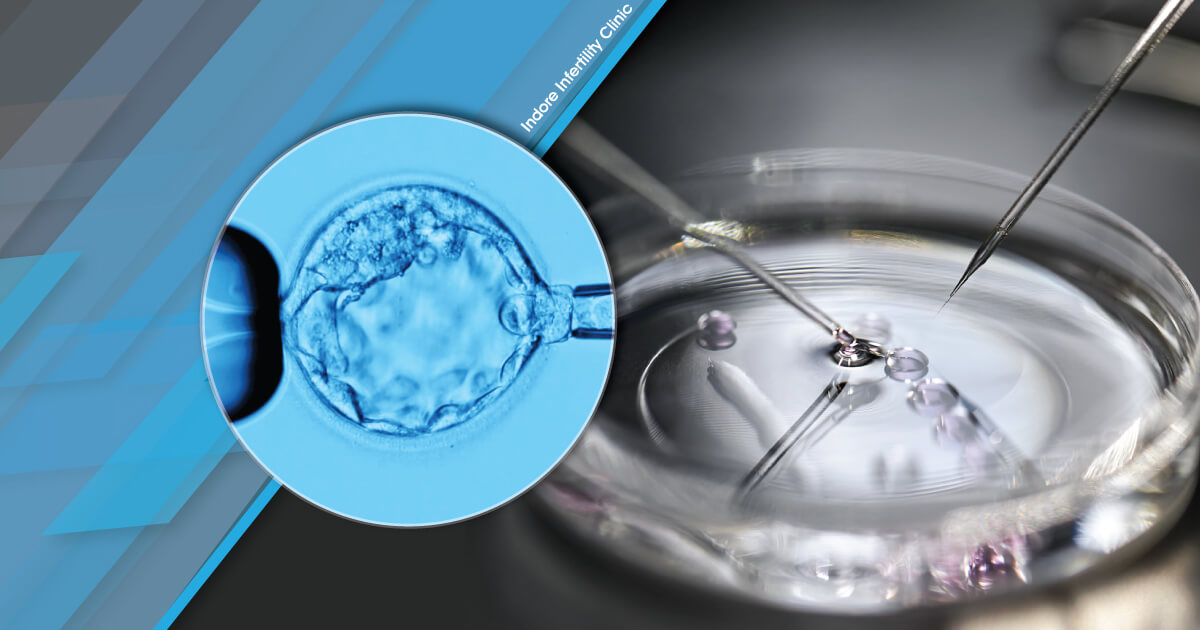
How PGT Works: The Science Behind Day 5 Testing
PGT-A (aneuploidy screening) analyzes 23 chromosome pairs in 5–10 trophectoderm cells (outer embryo cells) biopsied on Day 5. This timing is critical: by Day 5, the embryo has reached the blastocyst stage, where trophectoderm cells are genetically identical to the inner cell mass (which becomes the fetus) but can be safely removed without harming the embryo. The mechanism of action involves next-generation sequencing (NGS) to detect copy number variations (CNVs)—extra or missing chromosome segments—that cause disorders like Prader-Willi syndrome.
Key advances in 2026 include:
- Non-invasive PGT (niPGT): Emerging techniques analyze maternal blood or spent embryo culture media to avoid biopsy risks, though accuracy lags at ~85% [NEJM, 2025].
- Polygenic risk scoring: PGT now evaluates hundreds of genes linked to complex traits (e.g., Alzheimer’s), not just single-gene disorders.
- Mitochondrial replacement therapy (MRT): Approved in the UK (2024) for maternal mitochondrial DNA disorders, but banned in the US due to ethical concerns [FDA, 2023].
| Test Type | Disorders Detected | Accuracy (%) | Cost (USD) | Regulatory Status (2026) |
|---|---|---|---|---|
| PGT-A (Aneuploidy) | Down syndrome, Trisomy 18/13 | 99% | $3,500–$6,000 | FDA-approved (US), EMA-approved (EU) |
| PGT-M (Monogenic) | Cystic fibrosis, sickle cell | 99.8% | $5,000–$10,000 | FDA-approved (US), EMA-approved (EU) |
| PGT-SR (Structural Rearrangements) | Translocations, inversions | 98% | $7,000–$12,000 | Investigational (US), Conditional (EU) |
Global Disparities: Who Gets Access?
The geographical divide is stark:
- United States: PGT is widely available but not covered by Medicare or most private insurers. A 2025 study found only 12% of IVF cycles included PGT due to cost [JAMA, 2025].
- Europe: The EMA’s 2024 guidance mandates PGT counseling but leaves pricing to member states. Germany covers PGT for severe disorders, while the UK’s NHS offers it only in high-risk families.
- India: PGT is legal but unregulated. Private clinics charge $8,000–$15,000 per cycle, pricing out 90% of the population. The Indian Council of Medical Research (ICMR) has no national screening program for genetic disorders.
- Low-income countries: 0% access. A 2026 WHO report highlights that 95% of newborns in sub-Saharan Africa lack any genetic screening.
—Dr. Margaret Harris, WHO Director of Maternal and Child Health
“PGT is a game-changer for high-income couples, but it’s a luxury for the global majority. We need universal newborn screening programs—not just for IVF patients—to address the root of this crisis. The data is clear: 80% of genetic disorders are preventable with early detection.”
Funding and Bias: Who Pays—and Why?
The $1.2 billion global PGT market is dominated by:
- Genetic testing labs: Illumina (US) and Thermo Fisher (US) supply NGS platforms, with no-profit disclosure on pricing.
- IVF clinics: Reproductive Medicine Associates (RMA) in the US charges $25,000+ for PGT-IVF bundles, creating conflicts of interest.
- Pharma: Companies like Genentech fund research into gene therapies (e.g., for spinal muscular atrophy), but not screening infrastructure.
Public funding is scarce:
- The US NIH allocated $15 million in 2025 for genetic disorder research—but none for PGT access programs.
- The UK NHS spends £50 million/year on rare disease treatments but £0 on preventive screening.
- India’s Department of Biotechnology has no budget for PGT, despite 1 in 20 births affected by genetic disorders [NCBI, 2024].
Contraindications & When to Consult a Doctor
Who should not pursue PGT:
- Couples not undergoing IVF: PGT requires in-vitro fertilization; natural conception carries no screening option.
- Families with de novo (new) mutations: Some disorders (e.g., achondroplasia) arise spontaneously and can’t be predicted.
- Those with mitochondrial DNA disorders: PGT cannot correct mitochondrial defects; MRT is the only option (available only in the UK).
- Financial barriers: Without insurance coverage, the $10K–$20K cost may force difficult ethical choices.
When to seek medical advice:
- If you have a family history of genetic disorders (e.g., Huntington’s disease, hemophilia), discuss preconception carrier screening with a genetic counselor.
- If you’re over 35, the risk of aneuploidy (e.g., Down syndrome) rises; PGT-A may be recommended alongside IVF.
- If you’ve had multiple miscarriages or stillbirths, chromosomal abnormalities may be the cause—PGT-M could help.
- If you’re considering egg freezing, ask about pre-freeze genetic screening to avoid future PGT costs.
The Future: Can PGT Solve the Genetic Disorder Crisis?
The path forward requires:
- Policy: Mandate newborn screening for all births (not just IVF), as recommended by the CDC.
- Technology: Expand non-invasive PGT to eliminate biopsy risks, with 100% accuracy by 2030.
- Equity: Subsidize PGT in public IVF programs (e.g., India’s Assisted Reproductive Technology (ART) Bill 2026, pending).
- Education: Train 10,000+ genetic counselors globally to bridge the knowledge gap [WHO, 2026].
For now, PGT remains a tool for the privileged. But as costs drop and regulations align, it could become a cornerstone of preventive medicine. The question isn’t if genetic disorders will decline—it’s how quick we act.
References
- NEJM (2025): “Non-Invasive Preimplantation Genetic Testing: Accuracy and Ethical Considerations”
- JAMA (2025): “Barriers to PGT Access in the United States: A Cost-Effectiveness Analysis”
- WHO (2026): “Global Report on Genetic Disorders in Newborns: Gaps in Screening and Treatment”
- NCBI (2024): “Epidemiology of Genetic Disorders in South Asia: A Systematic Review”
- CDC (2026): “Recommended Uniform Screening Panel for Newborns”
Disclaimer: This article is for informational purposes only. Consult a licensed genetic counselor or fertility specialist for personalized medical advice.

