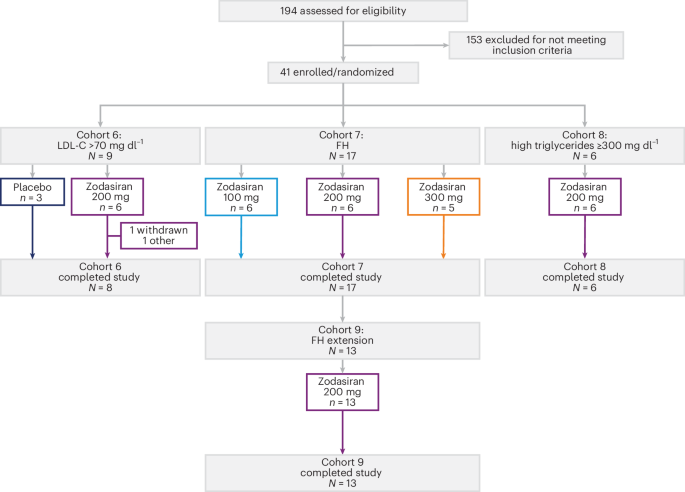
Zodasiran, a novel small interfering RNA (siRNA) therapy targeting the ANGPTL3 protein, has demonstrated significant efficacy in lowering triglycerides and LDL cholesterol in a Phase 1 basket trial published this week in Nature Medicine. The trial focuses on patients with severe hypertriglyceridemia and heterozygous familial hypercholesterolemia.
For millions of patients globally, managing lipids is not merely about diet and exercise; We see a battle against genetic predispositions. When the body cannot clear fats from the blood, the risk of acute pancreatitis and premature cardiovascular disease skyrockets. Zodasiran represents a shift toward “precision lipidology,” moving away from daily pills toward long-acting genetic silencers.
In Plain English: The Clinical Takeaway
- Genetic Silencing: Instead of managing symptoms, Zodasiran stops the production of a specific protein (ANGPTL3) that normally keeps fats in your bloodstream.
- Broad Impact: It works for two different high-risk groups: those with dangerously high triglycerides and those with a genetic form of high “bad” cholesterol (LDL).
- Long-Term Potential: Since it is an siRNA, this treatment may only be needed occasionally rather than every day, significantly reducing “pill fatigue.”
The Molecular Mechanism: How Zodasiran Silences ANGPTL3
To understand Zodasiran, we must understand the mechanism of action—the specific biochemical process through which a drug produces its effect. Zodasiran is a small interfering RNA (siRNA). Consider of siRNA as a “molecular eraser” that targets the messenger RNA (mRNA) before it can be translated into a protein.

The target here is Angiopoietin-like protein 3 (ANGPTL3). In a healthy metabolic system, ANGPTL3 acts as a brake, inhibiting the enzymes lipoprotein lipase (LPL) and endothelial lipase. By inhibiting these enzymes, ANGPTL3 keeps triglycerides and LDL cholesterol circulating in the blood.
By “silencing” the gene responsible for ANGPTL3, Zodasiran effectively removes the brake. This allows LPL to work unimpeded, rapidly breaking down triglycerides and clearing LDL from the plasma. This approach is fundamentally different from statins, which inhibit the production of cholesterol in the liver via the HMG-CoA reductase pathway.
Bridging the Gap: From Phase 1 Data to Global Patient Access
Whereas the Phase 1 “basket trial” (a study design that groups patients by a common molecular target regardless of their specific disease) proves safety and biological activity, the road to the clinic involves rigorous regulatory hurdles. In the United States, the FDA will require Phase 2 and 3 trials to prove that lowering these lipids actually reduces “major adverse cardiovascular events” (MACE), such as heart attacks or strokes.
In Europe, the European Medicines Agency (EMA) often emphasizes the benefit-risk ratio for orphan diseases, such as heterozygous familial hypercholesterolemia (HeFH). For patients in the UK, the NHS will eventually evaluate the cost-effectiveness of Zodasiran compared to existing therapies like PCSK9 inhibitors.
The funding for this research is primarily driven by biotechnology partnerships and pharmaceutical investment. Transparency in funding is critical because industry-sponsored trials can occasionally exhibit publication bias. However, the publication in Nature Medicine implies a rigorous peer-review process that validates the reported lipid reductions.
“The ability to target ANGPTL3 via siRNA provides a potent tool for patients who are refractory to traditional statin therapy, potentially altering the trajectory of cardiovascular risk for those with rare genetic lipid disorders.”
| Patient Cohort | Primary Target | Observed Effect | Clinical Significance |
|---|---|---|---|
| Severe Hypertriglyceridemia | Triglycerides | Significant Reduction | Lower risk of acute pancreatitis |
| Heterozygous Familial Hypercholesterolemia | LDL & Triglycerides | Dual Reduction | Reduced atherosclerotic plaque buildup |
Comparing siRNA to Traditional Lipid Therapies
The clinical landscape for hyperlipidemia has evolved. For decades, statins were the gold standard. Then came PCSK9 inhibitors—monoclonal antibodies that block the protein that degrades LDL receptors. While effective, these require frequent injections.
Zodasiran enters the “RNA interference” (RNAi) category. Unlike monoclonal antibodies, which bind to proteins already in circulation, siRNA prevents the protein from being created in the first place. This typically results in a longer duration of action, potentially reducing dosing frequency to once every six months.
According to data available via PubMed on similar RNAi therapies (like Inclisiran), the primary challenge is the delivery system. Zodasiran utilizes a GalNAc-conjugation, which ensures the drug is delivered specifically to the hepatocytes (liver cells), minimizing systemic side effects and reducing the risk of an immune response.
Contraindications & When to Consult a Doctor
While the Phase 1 results are promising, Zodasiran is not a universal cure. It is currently intended for patients with severe genetic lipid disorders, not for general “high cholesterol” caused by lifestyle factors.
Contraindications: Patients with severe hepatic impairment (liver failure) should avoid siRNA therapies targeting the liver, as the drug’s metabolism and delivery depend on functional hepatocytes. Those with a history of severe hypersensitivity to RNA-based therapeutics should exercise extreme caution.
When to seek immediate support: If you are participating in a clinical trial or using an approved lipid-lowering agent and experience unexplained muscle pain (myalgia), dark-colored urine, or yellowing of the skin (jaundice), consult your physician immediately. These can be signs of rhabdomyolysis or hepatotoxicity, though they are rare with the siRNA mechanism.
The Future of Precision Lipid Management
The final report of this Phase 1 basket trial confirms that Zodasiran is a potent modulator of lipid metabolism. However, the medical community must remain objective: a reduction in biomarkers (numbers on a blood test) is not the same as a reduction in mortality. The next three to five years will be critical as the drug moves into larger, double-blind placebo-controlled trials—the gold standard of medical evidence.
If Zodasiran maintains its safety profile and proves to reduce cardiovascular events, it will transition from a niche treatment for rare diseases to a cornerstone of public health intelligence in the fight against heart disease.
References
- Nature Medicine. (2026). Zodasiran for cholesterol and triglyceride lowering in patients with hyperlipidemia. doi:10.1038/s41591-026-04307-8
- World Health Organization (WHO). Cardiovascular diseases (CVDs) Fact Sheets. who.int
- Centers for Disease Control and Prevention (CDC). Heart Disease and Stroke Prevention. cdc.gov
- The Lancet. Lipid-lowering therapies and cardiovascular outcomes. thelancet.com
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.