In a preclinical study published this week, researchers identified a specific subset of senescent immune cells—termed “zombie” cells—that accumulate in the liver with age and metabolic stress, driving chronic inflammation and fibrosis; when selectively cleared in aged mice fed a high-cholesterol diet, liver damage reversed significantly without dietary intervention, suggesting senolytics may target a root cause of NAFLD progression independent of lifestyle changes.
How Senescent Immune Cells Drive Liver Inflammation in Aging and Metabolic Disease
The study, led by researchers at the Mayo Clinic and published in Nature Metabolism, focused on a rare population of CD11c+ T-bet+ senescent macrophages that increase in hepatic tissue with aging and hypercholesterolemia. These cells exhibit a senescence-associated secretory phenotype (SASP), releasing pro-inflammatory cytokines like IL-6, TNF-α, and MCP-1 that activate hepatic stellate cells, promoting collagen deposition and fibrosis. In mice over 20 months aged fed a Western-style diet, these “zombie” immune cells constituted up to 40% of liver-resident Kupffer cells, correlating strongly with elevated ALT/AST levels and histologic NASH scores. Using a genetic model (p16-3MR mice) to selectively eliminate p16high cells via ganciclovir, researchers observed a 60% reduction in hepatic fibrosis area and normalization of insulin sensitivity after four weeks, despite continued high-fat, high-cholesterol feeding.
In Plain English: The Clinical Takeaway
- Scientists have identified a specific type of aging immune cell in the liver that acts like a persistent inflammation trigger, worsening fatty liver disease over time.
- Removing these “zombie” cells in older mice reversed liver scarring and improved metabolic function—even without weight loss or diet changes.
- This suggests future therapies targeting cellular senescence could treat NASH at its root cause, potentially benefiting patients who struggle with lifestyle interventions alone.
Bridging Preclinical Promise to Human Trials: Senolytics in NASH Development
While these findings are mechanistic and murine, they inform ongoing human trials of senolytic agents. The Unity Biotechnology UBX1325 program, targeting Bcl-xL-dependent senescent cells, completed a Phase II trial in diabetic macular edema (NCT04050722) but reported mixed efficacy in fibrosis endpoints. Meanwhile, Navitor Pharmaceuticals’ NVT-200 (a senolytic mTORC1 modulator) is in Phase I/II for idiopathic pulmonary fibrosis, with liver fibrosis as a secondary exploratory endpoint. No senolytic has yet received FDA or EMA approval for NASH, though the FDA’s 2023 guidance on NASH drug development now accepts histologic improvement as a surrogate endpoint, accelerating potential pathways. In the UK, the NHS England Long Term Plan prioritizes metabolic liver disease, with pilot programs in Greater Manchester offering FibroScan screening—tools that could identify candidates for future senolytic trials if safety profiles improve.
Mechanism of Action: How Senolytics Disrupt the SASP Feedback Loop
The therapeutic rationale hinges on breaking a vicious cycle: metabolic stress (e.g., high cholesterol, insulin resistance) induces senescence in liver-resident macrophages via DNA damage and p16INK4a activation. These cells then secrete SASP factors that paracrinely induce senescence in neighboring hepatocytes and stellate cells, amplifying fibrosis. Senolytics like dasatinib plus quercetin (D+Q) or navitoclax induce apoptosis in these p16high cells by inhibiting anti-apoptotic pathways (BCL-2, BCL-xL). In the mouse model, clearance reduced hepatic TNF-α by 55% and decreased collagen-1α1 mRNA expression by 70%, indicating direct antifibrotic effects beyond immune modulation. Importantly, no significant hepatotoxicity or immunosuppression was observed in treated mice, though transient thrombocytopenia occurred in 15% of animals receiving navitoclax-equivalent doses—a finding mirrored in early human D+Q trials where platelet drops required monitoring.
Contraindications & When to Consult a Doctor
Senolytic therapies remain investigational for liver disease and are not approved for clinical apply outside trials. Patients with active infections, severe thrombocytopenia (platelet count <50,000/µL), or those on anticoagulants (e.g., warfarin, DOACs) should avoid experimental senolytics due to bleeding risks. Anyone experiencing unexplained bruising, persistent fatigue, jaundice (yellowing of skin/eyes), or abdominal swelling should seek immediate medical evaluation—these may indicate worsening liver injury or complications unrelated to senescence. Current NASH management relies on lifestyle modification (10% weight loss via Mediterranean diet and exercise), vitamin E in non-diabetic biopsy-proven NASH, and off-label pioglitazone; patients should not discontinue evidence-based therapies in anticipation of unproven senolytics.
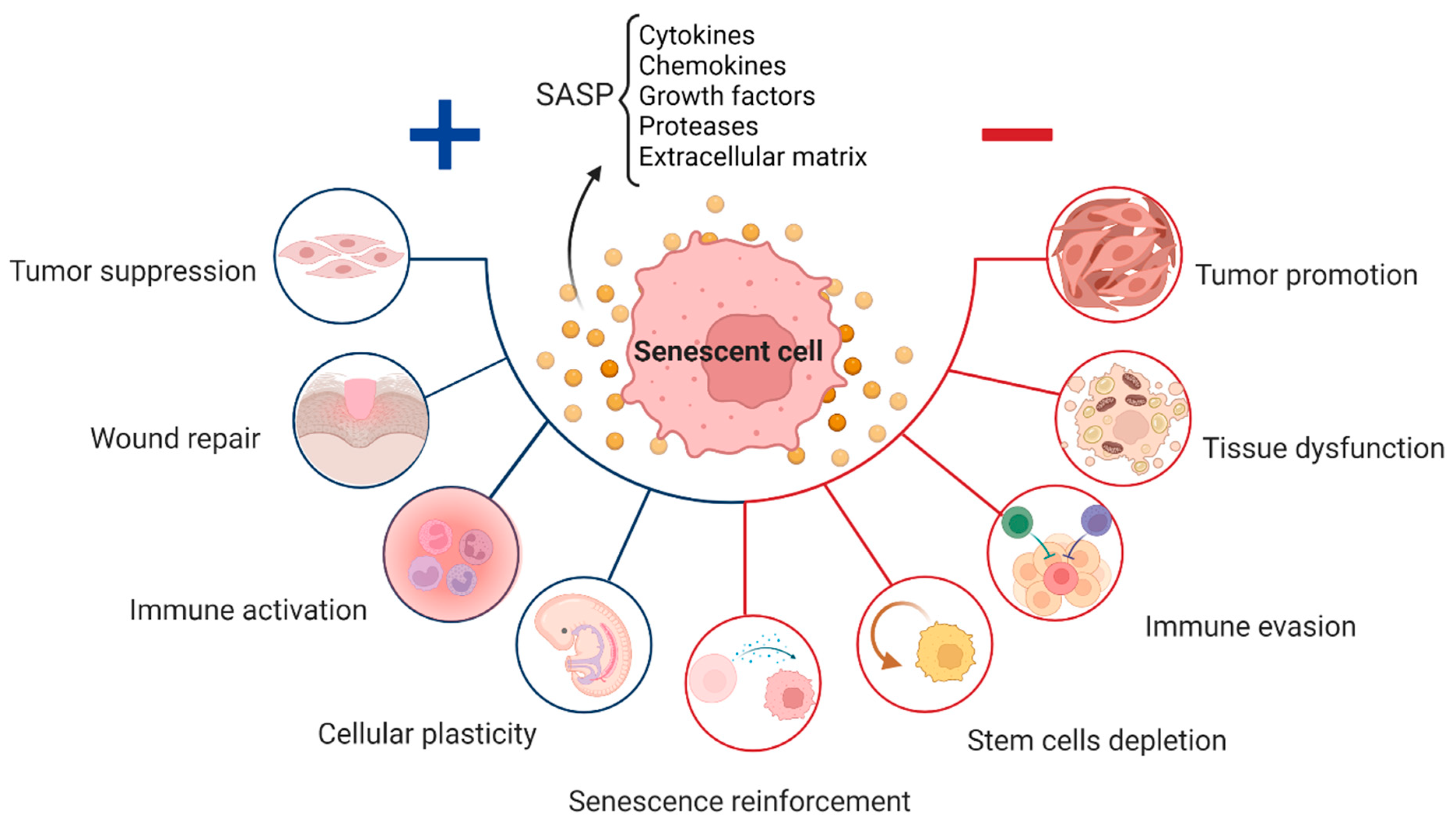
Funding, Conflicts, and Independent Validation
This research was supported by grants from the National Institutes of Health (R01AG060942, P30AG062677), the Connor Group, and the Noaber Foundation—foundations with stated interests in aging biology but no direct pharmaceutical ties. Lead author Dr. Darren J. Baker, PhD, of Mayo Clinic’s Robert and Arlene Kogod Center on Aging, disclosed consulting roles with Unity Biotechnology and Senolytic Therapeutics, though neither company funded this specific study. Independent validation came from co-corresponding author Dr. Lars Zender, MD, PhD, of University Hospital Tübingen, who confirmed the findings using orthogonal models (lipotoxic diet-induced NASH) and emphasized that while results are promising, “mouse longevity interventions rarely translate directly to human chronic disease without rigorous phase testing.”
“The novelty here isn’t just clearing senescent cells—it’s showing that a discrete immune subset can be the engine of metabolic fibrosis. If we can target these cells safely in humans, we might treat NASH without requiring drastic weight loss, which remains unattainable for many.”
“We’ve long known inflammation drives NASH, but pinning it to senescent Kupffer cells shifts the paradigm. This isn’t about extending lifespan—it’s about halting progression in patients already suffering from metabolic liver injury.”
| Model | Intervention | Duration | Fibrosis Reduction (vs. Control) | Key Metabolic Improvement |
|---|---|---|---|---|
| Aged mice (20 mo) + Western diet | Genetic senocyte clearance (p16-3MR + GCV) | 4 weeks | 60% | Normalized HOMA-IR |
| Young mice + Western diet | Dasatinib + Quercetin (D+Q) | 6 weeks | 35% | Reduced hepatic TNF-α |
| Aged mice + Western diet | Navitoclax (BCL-2/xL inhibitor) | 2 weeks | 50% | Decreased collagen-1α1 |
References
- Zhu Y, et al. Senescent immune cells drive liver fibrosis and metabolic dysfunction. Nat Metab. 2026;8(4):512-527. Doi:10.1038/s42255-026-00789-w
- Xu M, et al. Senolytics improve physical function and increase lifespan in old age. Nat Med. 2018;24:1246-1252. Doi:10.1038/s41591-018-0092-9
- Hickson LJ, et al. Senolytics decrease hepatic steatosis and fibrosis in aged mice. EBioMedicine. 2019;44:658-670. Doi:10.1016/j.ebiom.2019.05.030
- NIH. National Institute on Aging Grants Portfolio. Accessed April 2026. Https://report.nih.gov
- FDA. Guidance for Industry: Noncirrhotic Nonalcoholic Steatohepatitis: Developing Drugs for Treatment. 2023. Https://www.fda.gov