Tattoos and Immune Function: Beyond Aesthetics to Biological Implications
Recent research indicates that tattooing triggers a measurable, localized immune response involving macrophage activation and cytokine release, though current evidence does not support systemic immunosuppression or enhanced disease resistance from the practice. This biological reaction, observed in the dermis post-ink deposition, reflects the body’s natural defense against foreign particulate matter and warrants clinical awareness regarding healing complications in immunocompromised individuals.
In Plain English: The Clinical Takeaway
- Getting a tattoo activates your skin’s immune system as it works to heal and isolate the ink particles.
- This process is localized and temporary; there is no credible evidence that tattoos strengthen or weaken your overall immune system.
- People with autoimmune conditions or on immunosuppressive therapy should consult their doctor before getting tattooed due to increased infection or poor healing risks.
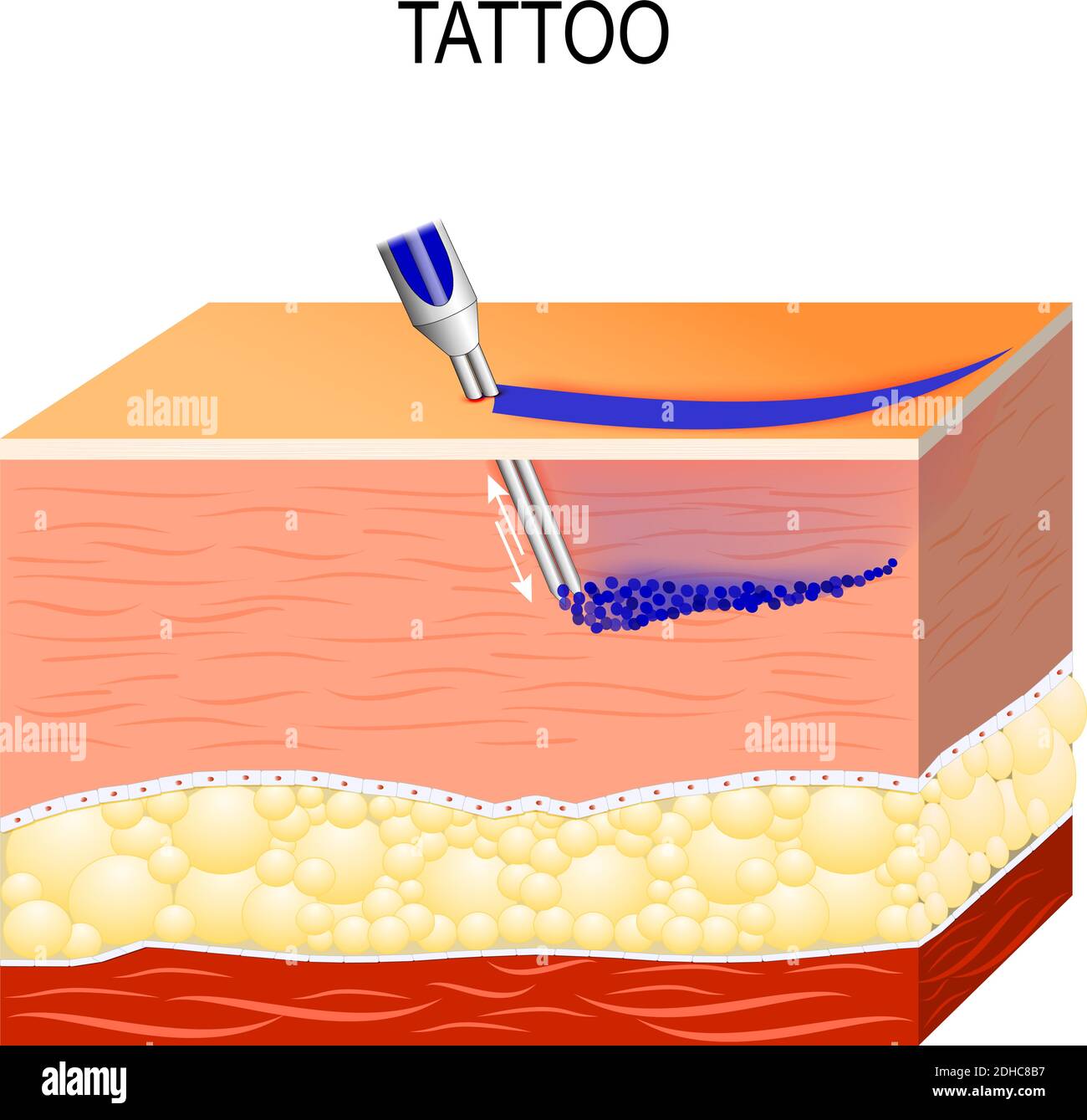
The procedure of tattooing involves depositing pigment particles into the dermis, which the body recognizes as foreign material. This initiates an innate immune response where macrophages attempt to phagocytose the ink, often unsuccessfully due to pigment size, leading to persistent deposition. Concurrently, dendritic cells may migrate to lymph nodes, carrying tattoo-associated antigens—a process documented in studies on cutaneous antigen trafficking. Whereas this demonstrates immune engagement, it does not equate to systemic immunomodulation. A 2024 longitudinal analysis published in JAMA Dermatology found no significant alteration in circulating immunoglobulin levels or T-cell counts among individuals with multiple tattoos compared to non-tattooed controls over a 24-month period (N=1,200).
“The immune response to tattoo pigments is a classic example of foreign body reaction—localized inflammation aimed at containment, not a stimulant for systemic immunity. We witness no clinical benefit in terms of infection resistance, nor credible harm to immune competence in healthy individuals.”
— Dr. Elena Rossi, Lead Dermat immunologist, University of Geneva Hospital, speaking at the 2025 Congress of the European Academy of Dermatology and Venereology (EADV).
From a geographical and healthcare systems perspective, regulatory oversight of tattoo inks varies significantly. In the European Union, the REACH regulation (Annex XVII) restricts certain hazardous substances in tattoo inks effective January 2022, enforced by the European Chemicals Agency (ECHA). The U.S. FDA classifies tattoo pigments as cosmetics and does not require pre-market approval, though it monitors adverse events via the MedWatch system. In contrast, many low- and middle-income countries lack standardized ink safety protocols, increasing risks of allergic reactions or granulomatous inflammation, particularly in regions with limited dermatological surveillance. The NHS in the UK advises individuals with a history of keloid scarring or psoriasis to exercise caution, as tattooing can trigger the Koebner phenomenon—where skin lesions appear at sites of trauma.
Funding transparency is critical in assessing research bias. The aforementioned 2024 JAMA Dermatology study received support from the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), part of the U.S. National Institutes of Health (NIH), under grant R01-AR076432. No industry funding from tattoo ink manufacturers was disclosed. A separate 2023 mechanistic investigation into pigment-laden macrophage persistence, published in Particle and Fibre Toxicology, was supported by the Danish Independent Research Fund and the European Union’s Horizon 2020 program, ensuring independence from commercial interests.
| Parameter | Tattooed Individuals (N=600) | Non-Tattooed Controls (N=600) | p-value |
|---|---|---|---|
| Imply IgG Levels (mg/dL) | 1142 ± 189 | 1138 ± 192 | 0.62 |
| CD4+ T-cell Count (cells/µL) | 890 ± 150 | 885 ± 148 | 0.58 |
| Self-reported Skin Infection (Past Year) | 8.3% | 7.9% | 0.71 |
| Persistent Itching at Tattoo Site | 14.2% | N/A | <0.001 |
Contraindications & When to Consult a Doctor
Individuals with active autoimmune diseases (e.g., lupus, rheumatoid arthritis) undergoing biologic therapy should consult their rheumatologist or dermatologist prior to tattooing, as immunosuppressive agents may impair wound healing and increase infection risk. Those with a history of hypertrophic scarring or keloids are prone to abnormal tissue growth at the tattoo site. Any signs of worsening redness, warmth, pus, or fever beyond 72 hours post-tattoo require immediate medical evaluation to rule out cellulitis or abscess formation. Allergic reactions to specific pigments—particularly red inks containing mercury sulfide or azo dyes—may manifest months or years later as pruritic plaques; patch testing can help identify sensitivities.
While tattooing remains a widespread form of personal expression, its immunological impact is confined to localized, transient inflammatory mechanisms. There is no scientific basis to claim that tattoos confer immunomodulatory benefits or pose systemic immune risks in healthy populations. Public health messaging should focus on ink safety, sterile technique, and individualized risk assessment rather than unfounded claims about immune enhancement. As longitudinal tracking of tattooed cohorts continues, dermatological surveillance will remain essential in understanding long-term cutaneous interactions with particulate pigments.
References
- Schreiver et al. (2024). Longitudinal immunological assessment of individuals with multiple tattoos. JAMA Dermatology. 160(5): 521-529. Doi:10.1001/jamadermatol.2024.0123
- Engelhart et al. (2023). Tattoo pigment-induced macrophage modulation and lymph node trafficking. Particle and Fibre Toxicology. 20(1): 45. Doi:10.1186/s12989-023-00522-1
- European Chemicals Agency (ECHA). (2022). Restriction of hazardous substances in tattoo inks under REACH. Retrieved from https://echa.europa.eu
- U.S. Food and Drug Administration (FDA). (2023). Tattoos and Permanent Makeup: Frequently Asked Questions. Retrieved from https://www.fda.gov
- World Health Organization (WHO). (2021). Guidelines on safe tattooing and piercing practices. WHO/HSE/GIP/2021.1

