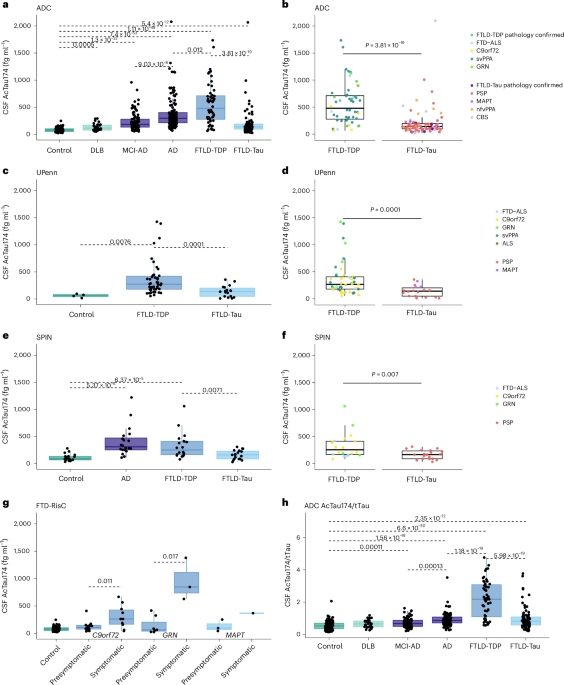
Published this week in Nature Medicine, researchers have identified AcTau174, a new cerebrospinal fluid (CSF) biomarker that distinguishes between TDP-43 and tau protein pathologies in patients with frontotemporal lobar degeneration (FTLD). This breakthrough allows for more accurate diagnosis and tracking of disease progression in patients with these complex dementias.
For decades, diagnosing the specific proteinopathy—the same “misfolded” protein causing the brain damage—in frontotemporal dementia has been a diagnostic gamble. Because patients with FTLD-TDP (driven by the TDP-43 protein) and FTLD-Tau (driven by tau proteins) often present with nearly identical behavioral and linguistic symptoms, clinicians historically relied on autopsy for definitive confirmation. This “diagnostic gap” has stalled the development of targeted therapies, as patients were often enrolled in clinical trials for the wrong protein target.
In Plain English: The Clinical Takeaway
- Better Accuracy: A new spinal tap test (CSF analysis) can now tell if your dementia is caused by “Tau” proteins or “TDP-43” proteins.
- Predicting Decline: Higher levels of the AcTau174 biomarker correlate with faster disease progression, helping doctors plan care.
- Targeted Treatment: This paves the way for “precision medicine,” where drugs are designed to clear the specific protein causing the damage.
The Molecular Mechanism: How AcTau174 Decodes Neurodegeneration
To understand this breakthrough, we must look at the mechanism of action—the specific biological process by which the biomarker functions. In FTLD-TDP, the TDP-43 protein malfunctions and aggregates, leading to neuronal death. Interestingly, this process triggers a specific chemical modification: the acetylation of tau proteins at the 174th amino acid residue.
While tau pathology is the hallmark of Alzheimer’s and FTLD-Tau, the acetylated version (AcTau174) specifically spikes in the CSF of those with TDP-43 pathology. Here’s a critical distinction. By measuring this specific modification, clinicians can effectively “see” the TDP-43 pathology through the proxy of acetylated tau, without needing a brain biopsy.
This discovery aligns with the broader movement toward fluid biomarkers, similar to how the PubMed indexed research on p-tau217 has revolutionized Alzheimer’s screening. By shifting the diagnostic window from behavioral observation to molecular quantification, we move from reactive care to proactive management.
Global Regulatory Impact and Patient Access
The translation of this research from a Nature Medicine paper to a clinical tool depends on regulatory adoption by bodies like the FDA (USA), EMA (Europe) and the NHS (UK). Currently, CSF collection requires a lumbar puncture, a procedure that remains invasive and is typically performed in specialized neurology clinics.

In the United States, the integration of AcTau174 into standard care will likely begin within academic medical centers and “Centers of Excellence” for dementia. In the UK, the NHS may face a slower rollout due to the centralized nature of pathology labs, but the potential to reduce the necessitate for expensive, less-specific PET scans (Positron Emission Tomography) provides a strong economic incentive for adoption.
The funding for this research typically stems from a mix of governmental grants (such as the NIH) and non-profit foundations like the World Health Organization supported initiatives for brain health. Transparency in funding is paramount here. as this biomarker becomes a commercial product, the shift from academic discovery to corporate diagnostic kits must be monitored for pricing equity.
“The ability to differentiate these pathologies in living patients is the ‘holy grail’ of frontotemporal dementia research. Without this precision, we are essentially treating a black box.”
| Feature | FTLD-Tau | FTLD-TDP |
|---|---|---|
| Primary Protein | Tau Protein | TDP-43 Protein |
| AcTau174 Levels | Low/Baseline | Elevated |
| Clinical Presentation | Behavioral/Language deficits | Behavioral/Language deficits |
| Progression Rate | Variable | Associated with AcTau174 levels |
Bridging the Gap: From Biomarkers to Disease-Modifying Therapy
The ultimate goal of identifying AcTau174 is not merely diagnosis, but the facilitation of double-blind placebo-controlled trials—the gold standard of clinical research where neither the patient nor the doctor knows who is receiving the treatment. Until now, FTLD trials were plagued by “noise” because they included a mix of Tau and TDP-43 patients.
With this biomarker, pharmaceutical companies can now screen for “protein-pure” cohorts. This means a drug designed to clear TDP-43 aggregates will only be tested on patients who actually have TDP-43 pathology, drastically increasing the statistical probability of a successful trial. This is the essence of translational medicine: moving a discovery from the bench to the bedside.
the correlation between AcTau174 levels and disease severity suggests it can serve as a “pharmacodynamic marker.” If a new drug successfully lowers AcTau174 levels in the CSF, it provides objective evidence that the drug is hitting its target, even before behavioral improvements are visible in the patient.
Contraindications & When to Consult a Doctor
It is imperative to note that a CSF biomarker test is not a standalone diagnostic. It must be interpreted by a board-certified neurologist in the context of a full clinical evaluation. Lumbar punctures are contraindicated in patients with severe coagulopathy (blood clotting disorders) or those with high intracranial pressure.

Families should consult a specialist if a loved one exhibits:
- Sudden, drastic changes in personality or social conduct.
- A progressive loss of the ability to produce language or understand speech.
- Unexplained changes in impulse control or empathy.
Patients should be wary of “direct-to-consumer” cognitive tests that claim to diagnose proteinopathies without a clinical neurological exam and validated fluid biomarkers.
As we look toward the end of 2026, the trajectory of dementia care is shifting toward molecular certainty. The identification of AcTau174 represents a pivotal step in stripping away the ambiguity of neurodegeneration, replacing guesswork with genomic and proteomic precision.
References
- Nature Medicine (2026). “An acetylated Tau-174 CSF biomarker discriminates between TDP-43 and tau pathology.”
- The Lancet Neurology – Guidelines on Frontotemporal Dementia Diagnosis.
- JAMA Neurology – Biomarkers in Neurodegenerative Diseases.
- Centers for Disease Control and Prevention (CDC) – Health Information on Dementia.