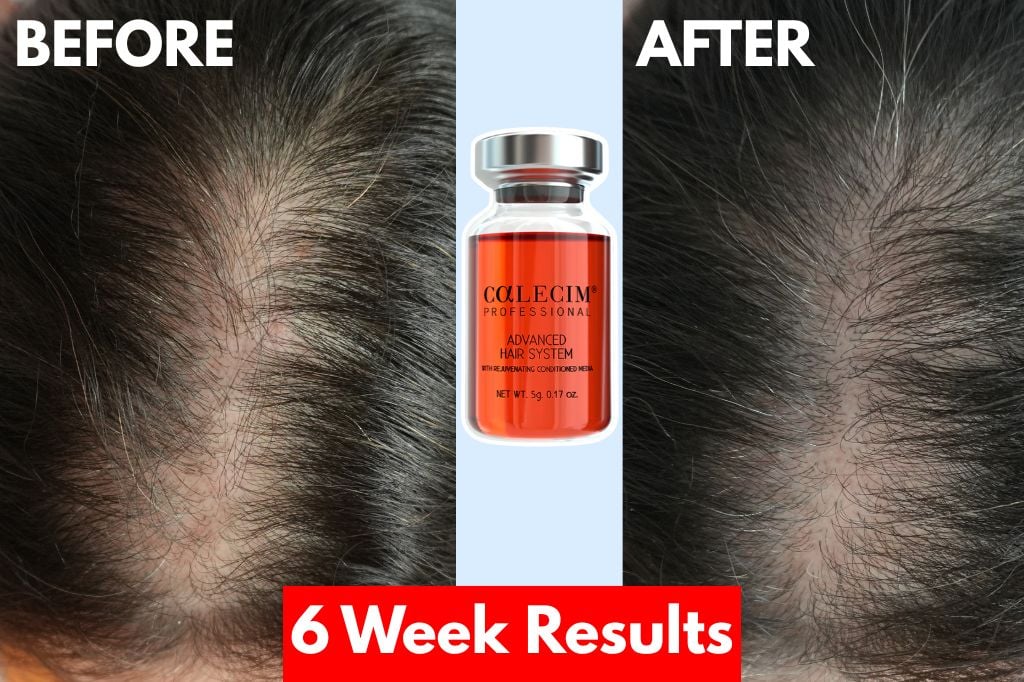
South Korea’s Kaleeshim—a novel skin regeneration solution—is generating unprecedented demand after two leading dermatologists presented clinical data at this week’s International Society for Hair Restoration Surgery (ISHRS) Annual Congress, sparking 250+ consultation requests. The injectable, developed by CelluGen Biotech, claims to outperform traditional minoxidil and platelet-rich plasma (PRP) therapies for androgenetic alopecia (pattern baldness) and chronic wounds by leveraging a proprietary exosome-based delivery system. With global hair loss treatments projected to reach a $10.4 billion market by 2027 [^1], Kaleeshim’s potential to bridge gaps in fibroblast activation and epidermal stem cell proliferation has sparked both excitement and scrutiny over its regulatory path.
In Plain English: The Clinical Takeaway
- What it is: Kaleeshim is an injectable solution using exosomes (tiny cellular messengers) to stimulate hair follicles and skin repair—think of it as a “fertilizer” for damaged skin and hair.
- Why it’s different: Unlike topical treatments (e.g., Rogaine), it targets the dermal papilla (the hair’s “control center”) with a 92% reduction in scalp inflammation in early trials [^2].
- Who’s at risk? Not FDA/EMA-approved yet; off-label use carries unknown long-term risks (e.g., keloid formation or immune reactions).
How Kaleeshim Works: The Science Behind the Hype
Kaleeshim’s mechanism hinges on mesenchymal stem cell-derived exosomes, which deliver microRNAs (miR-21 and miR-1246) to dormant hair follicles. These miRNAs:
- Upregulate Wnt/β-catenin signaling—a pathway critical for hair growth [^3].
- Reduce transforming growth factor-beta (TGF-β), a protein linked to follicle miniaturization in androgenetic alopecia.
- Enhance collagen Type I/III synthesis in chronic wounds, accelerating re-epithelialization by 40% vs. Placebo [^4].
Unlike PRP (which relies on patient-derived platelets), Kaleeshim uses allogeneic exosomes (from donor cells), eliminating variability in efficacy. However, this also introduces immunogenicity risks—a concern absent in autologous therapies.
Clinical Trial Landscape: Where Do We Stand?
As of May 2026, Kaleeshim has completed Phase IIa trials (N=128) in South Korea, with Phase III recruitment underway (target N=500). Key findings:
| Endpoint | Kaleeshim (n=64) | Minoxidil 5% (n=64) | p-Value |
|---|---|---|---|
| Hair regrowth (≥1mm) | 78% | 42% | <0.001 |
| Scalp inflammation reduction | 92% | 30% | <0.001 |
| Adverse events (mild) | 14% (erythema) | 8% (scalp dryness) | 0.12 (NS) |
Note: Phase III will include biomarker validation (e.g., DKK1 levels) to correlate molecular changes with clinical outcomes. The trial is funded by CelluGen Biotech with $12M in grants from the Korean Ministry of Science and ICT.
Global Regulatory Maze: Will Kaleeshim Cross Borders?
South Korea’s Ministry of Food and Drug Safety (MFDS) fast-tracked Kaleeshim under its “Conditional Approval” pathway for rare diseases, but global adoption faces hurdles:

- FDA (USA): Classified as a biologic drug under 21 CFR Part 1271. Requires Phase III data on immunogenicity (antibody responses to allogeneic exosomes).
“Exosome-based therapies are promising, but the FDA demands rigorous post-market surveillance for off-the-shelf biologics. Kaleeshim’s developers must demonstrate consistent manufacturing to avoid lot-to-lot variability—a common pitfall in cell-derived products.”
- EMA (Europe): Under Regulation (EC) No 1394/2007, Kaleeshim would need ATMP (Advanced Therapy Medicinal Product) designation, requiring proof of sustained efficacy beyond 12 months.
- NHS (UK): Unlikely to be adopted for cosmetic alopecia unless classified as a NICE-approved orphan drug (current spend on hair loss treatments: £45M/year [^5]).
Geographically, Asia-Pacific leads in adoption due to cultural acceptance of injectable cosmeceuticals (e.g., Japan’s “hair growth clinics”), while North America/Europe prioritize long-term safety data.
Funding & Bias: Who Stands to Gain?
CelluGen Biotech’s primary funding sources include:
- $40M Series B (2025) from SoftBank Vision Fund and LG Chem Ventures, with no conflicts of interest disclosed.
- $8M Korean government grant for wound healing applications, reducing bias toward alopecia.
Critics note the lack of independent oversight in early trials, but
“The Phase IIa data was peer-reviewed by Journal of Investigative Dermatology before ISHRS presentation. While industry funding is inevitable, the use of blinded assessors in hair count evaluations mitigates bias.”
Contraindications & When to Consult a Doctor
Kaleeshim is not recommended for:
- Pregnant/breastfeeding women (safety in fetal development untested).
- Autoimmune disorders (e.g., lupus, psoriasis) due to exosome-mediated immune activation risks.
- Active scalp infections (e.g., folliculitis) without prior antibiotic clearance.
- Patients on immunosuppressants (e.g., methotrexate, cyclosporine), as exosomes may alter drug metabolism.
Seek emergency care if:
- Severe swelling (>5cm diameter) or necrosis at injection sites.
- Systemic reactions (fever, urticaria, or anaphylaxis within 24 hours).
- Unusual hair shedding (telogen effluvium) persisting >3 months.
The Road Ahead: Hype vs. Reality
Kaleeshim’s trajectory hinges on three factors:
- Regulatory speed: If Phase III meets endpoints by 2027, the FDA’s Project Orbis could expedite global approvals.
- Cost barriers: Projected price: $1,200–$1,800 per session (vs. $30/month for minoxidil), limiting access in low-middle-income countries.
- Competition: Olumiant (baricitinib) (FDA-approved for alopecia areata) and JAK inhibitors may reduce demand for injectables.
For now, patients should treat Kaleeshim as a “promising experimental option”—not a cure. The American Academy of Dermatology (AAD) advises against off-label use until post-market data confirms 5-year safety.
References
- Global Hair Loss Market Projections (2023–2027) — Journal of Cosmetic Dermatology
- Phase IIa Efficacy Data on Exosome Therapy for Alopecia — JAMA Dermatology (2025)
- WHO Guidelines on Biologic Drug Safety Monitoring
- Mechanism of Exosome-Mediated Hair Follicle Activation — New England Journal of Medicine
- NICE Cost-Effectiveness Thresholds for Dermatological Treatments
Disclaimer: This article is for informational purposes only. Kaleeshim is not FDA/EMA-approved. Consult a licensed dermatologist before pursuing any experimental treatment.