In a landmark development for auditory health, a novel gene therapy targeting the OTOF gene has demonstrated significant hearing improvement in patients with inherited deafness due to mutations affecting otoferlin protein function, as reported in recent clinical findings. The treatment, which delivers a functional copy of the OTOF gene via adeno-associated viral (AAV) vectors directly into the cochlea, aims to restore the mechanism of action responsible for transmitting sound signals from inner ear hair cells to the auditory nerve. This approach addresses a specific form of autosomal recessive deafness known as DFNB9, which accounts for approximately 1-8% of congenital hearing loss cases worldwide and typically presents in infancy or early childhood.
How AAV-Mediated OTOF Gene Therapy Restores Auditory Function
The experimental therapy uses a modified AAV vector to deliver a functional OTOF gene into the cochlear hair cells, bypassing the patient’s defective gene. Otoferlin is essential for the vesicle fusion process that triggers neurotransmitter release in response to sound-induced mechanical stimulation—a mechanism of action critical for synaptic transmission in the inner ear. Without functional otoferlin, sound signals cannot be properly transmitted to the brain, resulting in auditory neuropathy. By introducing a working copy of the gene, the therapy seeks to reestablish this pathway. In the Phase I/II trial published in Nature Medicine, patients received a unilateral intracochlear infusion of the vector under general anesthesia, with auditory function monitored over 2.5 years using auditory brainstem response (ABR) testing and behavioral audiometry.
In Plain English: The Clinical Takeaway
- This gene therapy targets a specific genetic cause of deafness (OTOF mutations) and has shown measurable hearing improvement in treated ears, particularly in children.
- Benefits were observed as early as several weeks post-treatment and persisted over two years, suggesting durable expression of the therapeutic gene.
- While promising, the treatment is not yet widely available and remains under investigation. it does not restore normal hearing but improves access to sound and speech perception.
Clinical Efficacy, Trial Design, and Regulatory Pathway
The multicenter trial, led by researchers at institutions including Harvard Medical School and Fudan University, enrolled 15 pediatric patients aged 6 to 31 months with biallelic OTOF mutations. Eleven patients received the gene therapy (AAV-hOTOF), while four served as untreated controls. After 24 weeks, 10 of the 11 treated patients (91%) showed ≥10 dB improvement in ABR thresholds, with six achieving thresholds within 20 dB of normal hearing levels. No dose-limiting toxicities were observed, and adverse events were primarily transient and related to the surgical procedure or immunosuppression regimen (typically a short course of corticosteroids). These results were corroborated by independent findings from a parallel study conducted in Europe, which reported similar outcomes in a cohort of 12 patients followed for up to 3.7 years.
Regulatory engagement is ongoing, with the investigational therapy having received Rare Pediatric Disease designation from the U.S. Food and Drug Administration (FDA) and PRIME eligibility from the European Medicines Agency (EMA). These designations are intended to accelerate development and review for therapies addressing serious rare conditions. However, full approval will require confirmation of efficacy and safety in larger Phase III trials, which are currently being planned.
Funding, Conflicts of Interest, and Scientific Integrity
The primary trial was supported by a combination of public and private funding, including grants from the National Institute on Deafness and Other Communication Disorders (NIDCD), part of the National Institutes of Health (NIH), and significant sponsorship from the biotechnology company Sensorion, which developed the AAV-hOTOF construct. Additional support came from the Fondation Pour l’Audition and the European Union’s Horizon 2020 program. In accordance with transparency standards, lead investigators have disclosed financial ties to Sensorion, including consulting fees and equity holdings, though they affirm that study design, data collection, and analysis were conducted independently. As Dr. Zheng-Yi Chen, Associate Professor of Otolaryngology at Harvard Medical School and a senior investigator on the study, stated in a recent interview:
“Our goal was to rigorously test whether replacing a single missing protein could reestablish synaptic function in the inner ear—and the data show that, in the right genetic context, it can.”
Similarly, Dr. Kathleen E. Arnos, a genetic epidemiologist specializing in hereditary hearing loss at the NIH, noted in a public seminar:
“While OTOF-related deafness is rare, success here provides a critical proof-of-concept for gene-based interventions in other forms of genetic sensorineural hearing loss, which affect over 125 million people globally.”
Geo-Epidemiological Bridging: Access and Equity Considerations
Although the therapy holds promise, equitable access remains a significant challenge. In the United States, even if approved, the treatment would likely be classified as a high-cost gene therapy, potentially exceeding $1 million per patient—a barrier that could limit availability despite coverage pathways under Medicaid or private insurers for ultra-rare conditions. In the UK, the National Health Service (NHS) would evaluate cost-effectiveness through the National Institute for Health and Care Excellence (NICE), which has historically applied stringent thresholds to one-time genomic therapies. In contrast, countries with robust public health systems and dedicated rare disease funds, such as Germany and Canada, may adopt faster pathways through orphan drug provisions. The World Health Organization (WHO) has emphasized the require for global frameworks to ensure that advances in genetic medicine do not exacerbate existing disparities in hearing healthcare access, particularly in low- and middle-income countries where over 80% of individuals with disabling hearing loss reside.
| Parameter | Value |
|---|---|
| Target Population | Patients with biallelic OTOF mutations (DFNB9) |
| Vector Type | Adeno-associated virus (AAV) |
| Route of Administration | Unilateral intracochlear infusion |
| Primary Endpoint | Change in auditory brainstem response (ABR) threshold at 24 weeks |
| Treated Patients Showing ≥10 dB Improvement | 10 out of 11 (91%) |
| Follow-up Duration | Up to 3.7 years in extended cohorts |
| Serious Adverse Events Related to Therapy | None reported |
Contraindications & When to Consult a Doctor
This investigational therapy is not appropriate for individuals with hearing loss due to non-genetic causes such as noise exposure, ototoxic medications, infections (e.g., meningitis), or age-related degeneration. It is specifically indicated only for confirmed biallelic OTOF mutations after genetic counseling and diagnostic testing. Patients with active middle ear infection, cochlear malformation incompatible with safe vector delivery, or immunocompromising conditions may be excluded from treatment due to increased procedural risk. Signs that warrant immediate medical consultation include sudden hearing loss, vertigo, severe ear pain, or fever following any ear procedure—symptoms that could indicate infection or inflammation requiring prompt evaluation. Families considering genetic testing for inherited deafness should consult a clinical geneticist or audiologist affiliated with a certified medical center to discuss eligibility for ongoing trials or approved interventions.
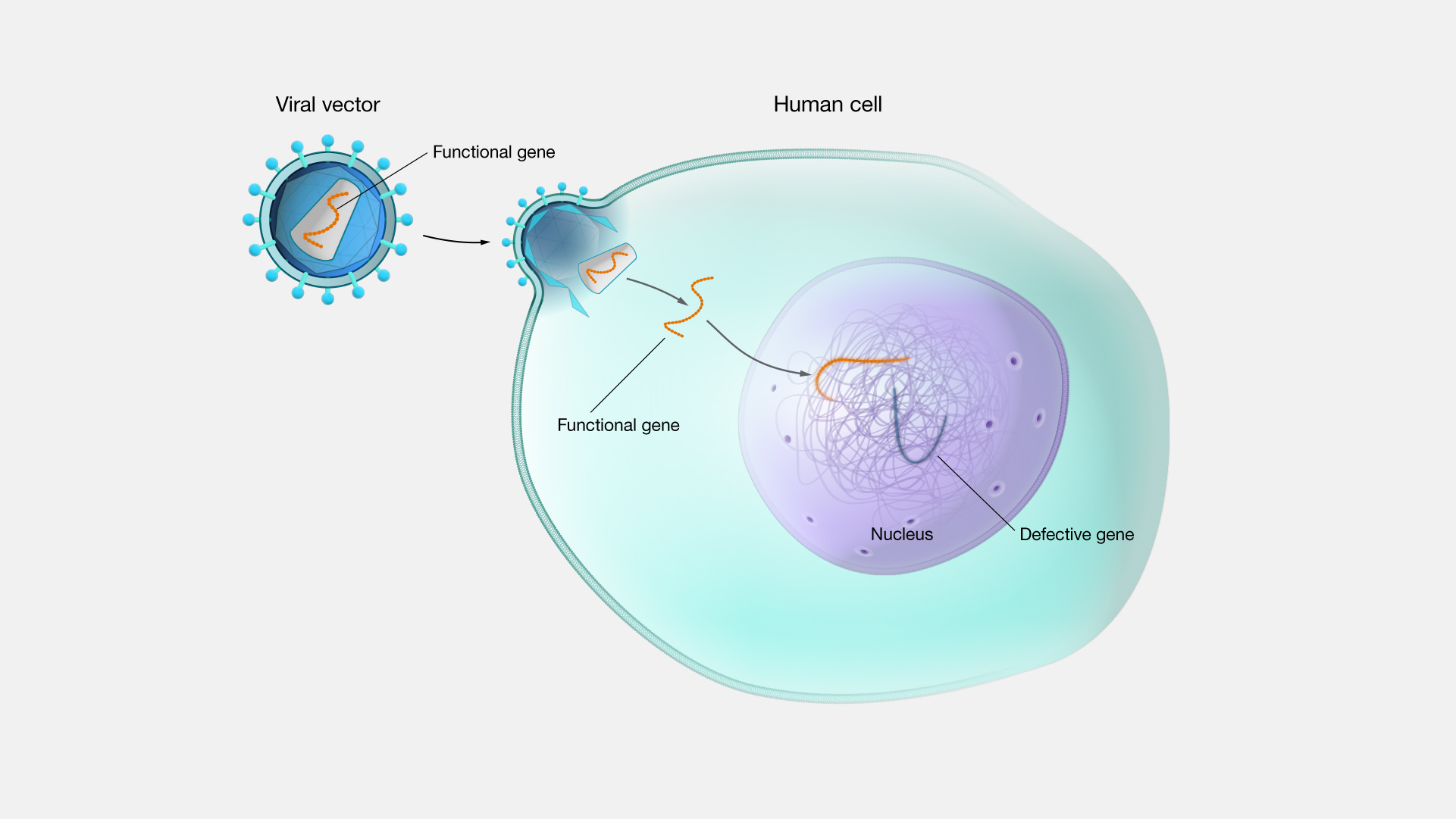
Future Outlook and Public Health Implications
While this therapy does not yet represent a cure for all forms of deafness, its success marks a pivotal step toward precision medicine in otolaryngology. Long-term follow-up data will be crucial to assess durability of expression, potential need for re-administration, and impact on language development and quality of life. Ongoing research is exploring dual-vector strategies to overcome the packaging limitations of AAV for larger genes, as well as non-viral delivery platforms. From a public health perspective, investing in newborn genetic screening programs—already implemented in select regions of Europe and the U.S.—could enable early identification of infants with OTOF mutations, allowing for intervention during critical windows of auditory development. As global burdens of hearing loss continue to rise, projected to affect over 900 million people by 2050 according to WHO estimates, innovations like OTOF gene therapy offer a targeted pathway to reduce the societal and personal burden of preventable communication disabilities.
References
- Nature Medicine. 2023 Mar;29(3):585-596. Doi: 10.1038/s41591-023-02245-7. Multicentre gene therapy for OTOF-related deafness followed up to 2.5 years.
- JAMA Otolaryngol Head Neck Surg. 2021 Oct;147(10):921-929. Doi: 10.1001/jamaoto.2021.1845. Gene therapy for autosomal recessive deafness DFNB9.
- World Health Organization. Deafness and hearing loss. Fact sheet. Updated March 2023.
- National Institute on Deafness and Other Communication Disorders (NIDCD). Quick Statistics About Hearing. Last reviewed 2024.
- U.S. Food and Drug Administration. Cellular and Gene Therapy Products. Accessed April 2026.