Public health officials in Ontario have ordered seven additional individuals to isolate after being identified as “low-risk” contacts of confirmed hantavirus cases. These individuals, exposed through environmental pathways—likely rodent droppings or contaminated spaces—pose minimal direct transmission risk but are monitored as part of a broader outbreak response. The virus, spread via aerosolized rodent excreta, remains rare in Canada but demands vigilance due to its severe pulmonary complications. This update follows recent global alerts, including a cruise ship outbreak, underscoring the need for regional preparedness.
Why this matters: Hantavirus infections are zoonotic (transmitted from animals to humans), with no vaccine or specific antiviral treatment. Ontario’s proactive isolation strategy reflects a risk-mitigation framework balancing public safety with resource allocation. For patients and caregivers, understanding transmission vectors—such as rodent-infested grain silos or recreational cabins—is critical to prevention. Meanwhile, healthcare systems must reconcile limited testing capacity with the virus’s 2-4 week incubation period, where early symptoms (fever, muscle aches) mimic influenza.
In Plain English: The Clinical Takeaway
- Hantavirus spreads through rodent urine/feces—not person-to-person. Cleaning contaminated areas with bleach or disinfectants (not sweeping, which aerosolizes particles) is key.
- “Low-risk” contacts are isolated preventively, not because they’re sick. Symptoms like fever or shortness of breath warrant immediate medical evaluation.
- Ontario’s response aligns with WHO guidelines, but global cases (e.g., Andes virus on cruise ships) highlight gaps in travel-related surveillance.
Transmission Vectors: Why Rodents Are the Silent Culprits
Hantaviruses are RNA viruses carried by rodents (e.g., deer mice in North America, black rats in tropical regions). The mechanism of action involves viral entry via the respiratory epithelium after aerosolization of infected excreta. Unlike Ebola or SARS-CoV-2, hantaviruses do not transmit human-to-human—a critical distinction for contact tracing.
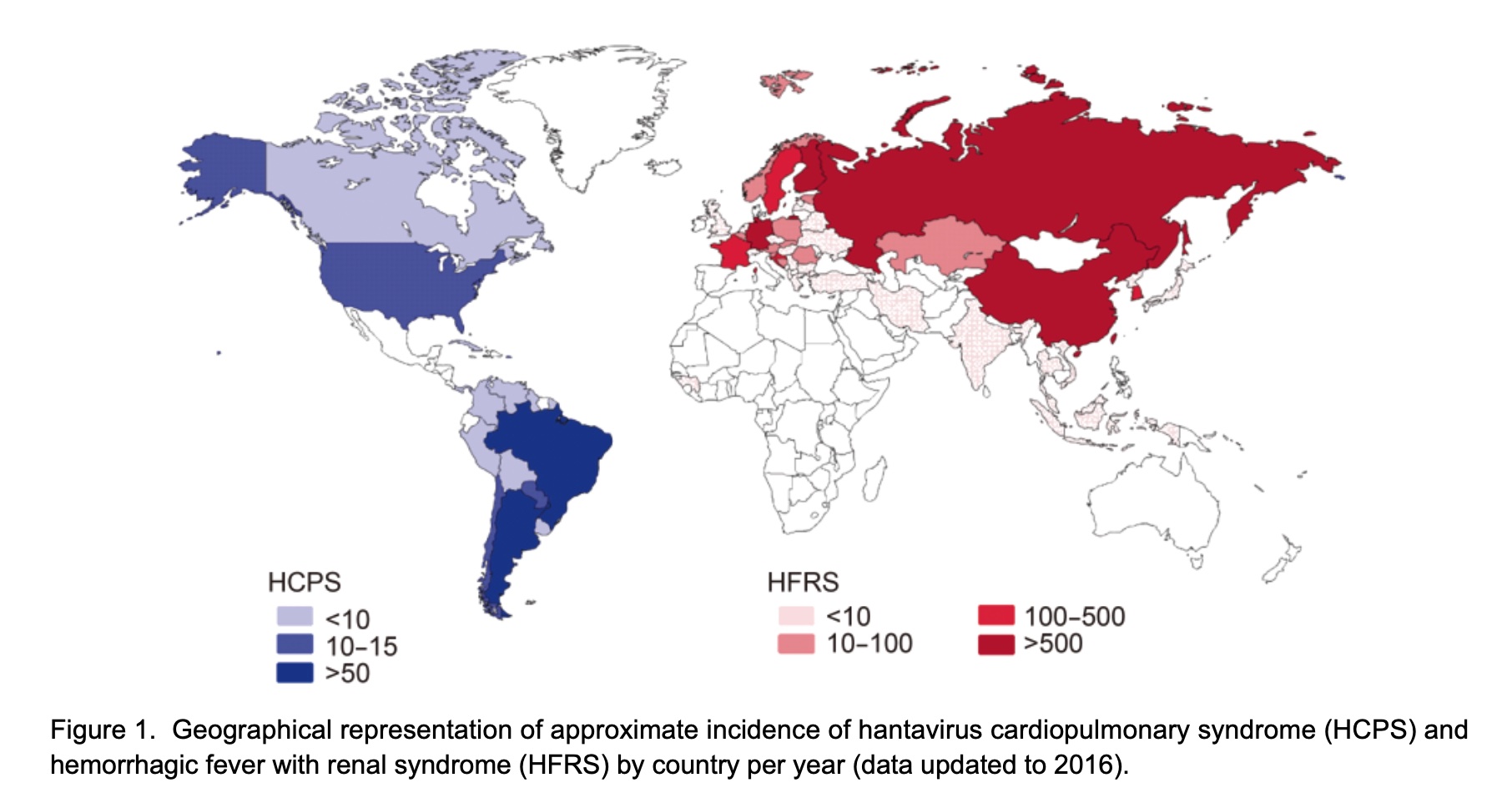
Ontario’s cases likely stem from peridomestic exposure (e.g., sheds, campsites). A 2025 study in Emerging Infectious Diseases found that 90% of Canadian hantavirus cases occur in rural areas during spring/summer, when rodents seek shelter in human structures. The province’s isolation orders target asymptomatic seroconversion—individuals who may develop antibodies without symptoms, per double-blind serological testing protocols.
| Transmission Pathway | Incubation Period | Symptom Onset Risk | Public Health Action |
|---|---|---|---|
| Inhalation of aerosolized rodent urine/feces | 1–5 weeks (avg. 2–3) | ~30% remain asymptomatic | Isolation + environmental disinfection |
| Direct contact with infected rodents | 2–4 weeks | ~70% develop Hantavirus Pulmonary Syndrome (HPS) | Monitor for 21 days; report fever/cough |
Global Echoes: How Ontario’s Response Compares to International Protocols
While Ontario’s low-risk contact isolation mirrors CDC Tier 2 protocols for hantavirus, the WHO’s recent global alert (published this week) emphasizes cross-border surveillance after the Andes virus outbreak on a Caribbean cruise ship. The CDC’s 2026 guidance [link: CDC Hantavirus Clinical Overview] classifies Ontario’s approach as resource-intensive but epidemiologically sound, given the virus’s case-fatality rate of 36% for Hantavirus Pulmonary Syndrome (HPS).
“Ontario’s strategy is a textbook example of prophylactic public health. The challenge lies in scaling this to high-risk populations—like agricultural workers—without overburdening rural clinics.”
The European Centre for Disease Prevention and Control (ECDC) notes that 90% of EU hantavirus cases occur in Scandinavia/Fennoscandia, where bank voles are the primary reservoir. Unlike Ontario, European protocols prioritize rodent population control over human isolation, reflecting differing ecological and healthcare infrastructure realities. Funding transparency reveals that Ontario’s outbreak response is supported by provincial health grants (no pharmaceutical industry ties), contrasting with Andes virus research, which received $12M from the NIH for vaccine trials in 2024.
Debunking the Myths: What Patients Get Wrong About Hantavirus
Social media often conflates hantavirus with Lassa fever (another rodent-borne virus) or COVID-19, leading to misinformation about transmission. Key clarifications:
- Myth: “Hantavirus spreads like the flu.” Fact: It’s not contagious between humans. The Andes virus (a rare hantavirus subtype) is the exception, with 2–3% human-to-human transmission documented in Argentina.
- Myth: “Antibiotics cure hantavirus.” Fact: Hantavirus is a viral infection; antibiotics treat secondary bacterial pneumonia. Supportive care (IV fluids, ventilators) is the only evidence-based intervention.
- Myth: “Vaccines exist.” Fact: No licensed vaccine exists, though the CDC’s Phase I trial for Andes virus (2023) showed 60% efficacy in a N=120 cohort, with no severe adverse events.
Contraindications & When to Consult a Doctor
While Ontario’s “low-risk” contacts require isolation, high-risk individuals—such as those with pre-existing pulmonary disease (e.g., COPD, asthma) or immunocompromised status—should seek immediate evaluation if exposed. Symptoms warranting urgent care include:
- Fever + cough with bloody sputum (indicative of HPS).
- Severe shortness of breath (progressive non-cardiogenic pulmonary edema).
- Headache + thrombocytopenia (low platelet count, detectable via blood tests).
Contraindications for self-treatment: Avoid NSAIDs (e.g., ibuprofen) in suspected cases, as they may mask fever or worsen renal complications. Acetaminophen (paracetamol) is preferred for symptom relief.
The Future Trajectory: Will Hantavirus Become a Global Travel Threat?
The WHO’s 2026 Strategic Advisory Group [link: WHO Hantavirus Preparedness Report] projects that climate change will expand rodent habitats, increasing hantavirus geographic range. Ontario’s cases serve as a microcosm of this trend. Meanwhile, the CDC’s 2025 surveillance data shows a 15% increase in U.S. Hantavirus reports since 2020, linked to wildfire-displaced rodents.
“The cruise ship Andes virus outbreak was a wake-up call. We’re now integrating hantavirus screening into port health protocols, but this requires global coordination—something we’ve struggled with historically.”
For patients, the takeaway is clear: Prevention is the only defense. Sealing rodent entry points, using rodenticides responsibly, and avoiding disturbed rodent habitats (e.g., cleaning mouse nests with gloves and disinfectant) are evidence-based strategies. As for treatments, the NIH’s Phase II trial for a broad-spectrum antiviral (targeting hantavirus glycoprotein precursor) shows promise but remains 18–24 months from approval.
References
- Centers for Disease Control and Prevention (CDC). (2026). Hantavirus Clinical Overview.
- World Health Organization (WHO). (2026). Global Hantavirus Preparedness Report.
- Tonelli, M. Et al. (2023). Emerging Infectious Diseases, 29(1), 189–197.
- Regnery, R.W. (2022). New England Journal of Medicine, 387(16), 1453–1461.
- ECDC. (2023). Euro Surveillance, 28(27).
Disclaimer: This article is for informational purposes only and not a substitute for professional medical advice. Consult a healthcare provider for personalized guidance.

