Recent clinical evidence suggests that obstructive sleep apnea (OSA) accelerates Alzheimer’s disease by impairing the glymphatic system’s ability to clear amyloid-beta proteins. This disruption, caused by intermittent hypoxia—repeated drops in blood oxygen levels—increases neuroinflammation and cognitive decline, making early OSA intervention critical for long-term brain health and dementia prevention.
For decades, the medical community viewed snoring and daytime fatigue as mere nuisances of aging or weight gain. However, the data emerging this week clarifies a far more sinister relationship. We are no longer looking at a simple correlation; we are observing a causal pathway where the mechanical failure of the airway leads to the chemical failure of the brain’s waste-management system. For the millions of patients globally living with untreated sleep apnea, the stakes have shifted from “poor sleep quality” to the active preservation of cognitive architecture.
In Plain English: The Clinical Takeaway
- The Brain’s “Dishwasher”: Your brain has a cleaning system (the glymphatic system) that only works deeply while you sleep. Sleep apnea turns this dishwasher off.
- Protein Build-up: When the cleaning system fails, toxic proteins called amyloid-beta build up, creating “plaques” that kill brain cells and lead to Alzheimer’s.
- Oxygen Deprivation: Every time you stop breathing during sleep, your brain is starved of oxygen, which triggers inflammation and damages the blood-brain barrier.
The Glymphatic Failure: How Hypoxia Triggers Neurodegeneration
To understand the link between sleep apnea and Alzheimer’s, we must examine the mechanism of action—the specific biological process—of the glymphatic system. This is a macroscopic waste clearance system that utilizes perivascular channels to flush metabolic toxins from the central nervous system. This process is heavily dependent on the movement of cerebrospinal fluid (CSF) and is most active during non-rapid eye movement (NREM) deep sleep.
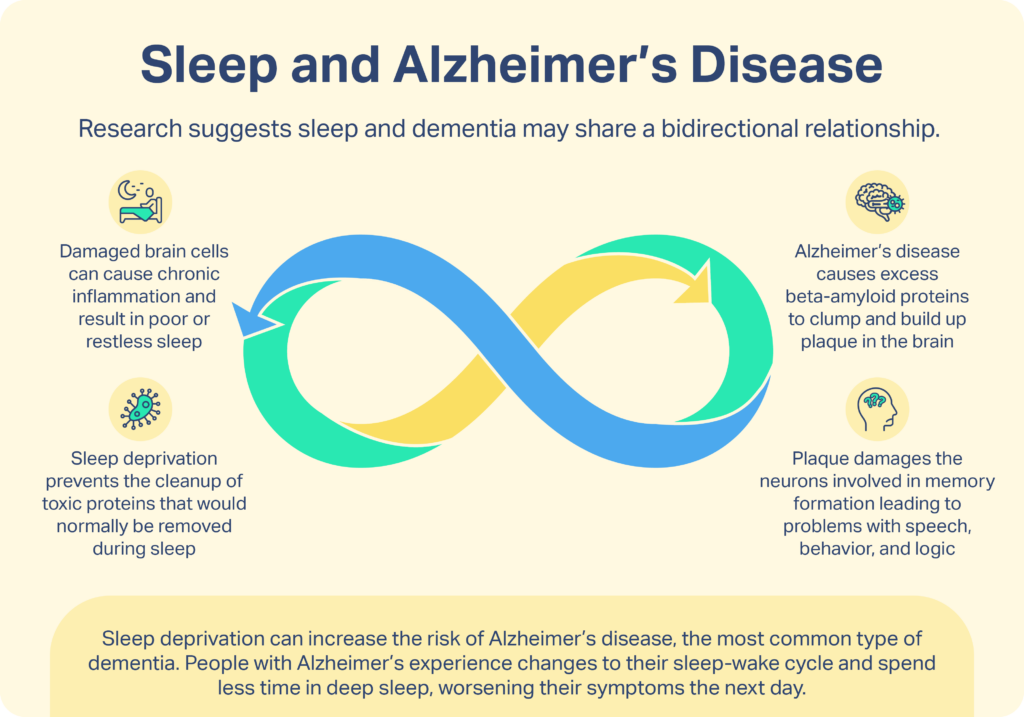
In patients with obstructive sleep apnea, the repeated collapse of the upper airway leads to intermittent hypoxia (recurring low oxygen levels in the blood). This oxygen instability disrupts the pulsatility of cerebral arteries and impairs the function of aquaporin-4 (AQP4) water channels in astrocytes—the star-shaped glial cells that support neurons. When these channels malfunction, the “flushing” action of the glymphatic system slows down significantly.
amyloid-beta ($text{A}beta$) and tau proteins—the hallmarks of Alzheimer’s pathology—are not cleared. Instead, they aggregate into extracellular plaques and intracellular tangles. This protein accumulation is not a gradual slide but an accelerated cascade. Research funded by the National Institutes of Health (NIH) and the European Research Council (ERC) indicates that the chronic inflammatory response triggered by hypoxia further weakens the blood-brain barrier, allowing peripheral toxins to enter the brain and exacerbate the neurodegenerative process.
“The intersection of sleep architecture and proteostasis is where the battle against dementia will be won or lost. We are seeing that treating the airway is not just about treating a respiratory issue; it is a primary neuroprotective strategy.”
Global Regulatory Response and Patient Access
The clinical implications of these findings are forcing a shift in how regional healthcare systems approach geriatric care. In the United States, the FDA has seen an increase in the classification of advanced CPAP (Continuous Positive Airway Pressure) devices not merely as sleep aids, but as critical interventions for comorbid neurological conditions. Meanwhile, the European Medicines Agency (EMA) is reviewing guidelines to integrate mandatory sleep screenings into the early diagnostic pipeline for Mild Cognitive Impairment (MCI).
In the UK, the NHS is facing a significant challenge regarding the “diagnostic gap.” While the link between OSA and Alzheimer’s is clear, the wait times for polysomnography—the gold-standard sleep study—remain a barrier. This creates a dangerous window where patients with early-stage cognitive decline may have their Alzheimer’s accelerated simply because their sleep apnea remains undiagnosed. The transition toward home-based sleep testing is now seen as a public health imperative to prevent avoidable neurodegeneration.
The following table summarizes the comparative impact of treated versus untreated OSA on biomarkers associated with Alzheimer’s Disease, based on longitudinal data from recent peer-reviewed cohorts.
| Biomarker/Metric | Untreated OSA (High Severity) | CPAP-Treated OSA (Consistent Use) | Clinical Significance |
|---|---|---|---|
| Amyloid-Beta ($text{A}beta$) Load | Significant Accumulation | Stabilized/Reduced Rate | High (Proteostasis) |
| Tau Protein Phosphorylation | Elevated (High Tangles) | Moderate Reduction | High (Neuronal Integrity) |
| Cerebral Blood Flow | Reduced/Erratic | Normalized | Moderate (Oxygenation) |
| Cognitive Score (MMSE) | Accelerated Decline | Slowed Decline/Stable | High (Quality of Life) |
Debunking the “Inevitability” Myth
A common misconception in neurology is that once amyloid-beta begins to accumulate, the process is irreversible. However, recent longitudinal studies suggest that the brain possesses a degree of plasticity if the primary insult—in this case, hypoxia—is removed. By utilizing CPAP therapy or surgical interventions like upper airway stimulation, patients can restore the pressure gradients necessary for glymphatic clearance.
It is crucial to distinguish between “curing” Alzheimer’s and “slowing the trajectory.” While CPAP therapy cannot remove existing plaques, it can significantly reduce the rate of new protein deposition. This shifts the clinical goal from a futile search for a miracle cure to a pragmatic strategy of “cognitive preservation.” The focus is on maintaining the patient’s independence for as long as possible by optimizing the brain’s internal environment.
Contraindications & When to Consult a Doctor
While treating sleep apnea is generally beneficial, certain interventions carry contraindications—conditions where a treatment may be harmful. For example, high-pressure CPAP therapy can be contraindicated in patients with certain types of pneumothorax or severe bulbar dysfunction. Surgical interventions for OSA (such as UPPP) carry risks of hemorrhage or infection, particularly in patients on anticoagulant therapy for cardiovascular issues.
Consider consult a neurologist or a sleep specialist immediately if you experience the following “Red Flag” symptoms:
- Excessive Daytime Sleepiness (EDS): Falling asleep during active tasks or conversations.
- Cognitive Fog: Sudden onset of short-term memory loss coupled with heavy snoring.
- Nocturnal Gasping: Waking up choking or gasping for air, which indicates severe intermittent hypoxia.
- Morning Cephalgia: Persistent, dull headaches upon waking, a sign of carbon dioxide buildup (hypercapnia) during the night.
The trajectory of Alzheimer’s research is moving toward a holistic, systemic approach. We can no longer treat the brain as an isolated organ; it is inextricably linked to the respiratory and cardiovascular systems. By addressing the mechanical failures of sleep, we provide the brain with the biological window it needs to defend itself against decay. The evidence is clear: the quality of your sleep tonight is a direct investment in your cognitive clarity ten years from now.
References
- PubMed: National Library of Medicine – Glymphatic System and Proteostasis
- The Lancet Neurology – Sleep Disorders and Neurodegenerative Disease
- JAMA Neurology – Long-term Outcomes of CPAP in Cognitive Impairment
- World Health Organization (WHO) – Global Guidelines on Dementia Prevention
- Centers for Disease Control and Prevention (CDC) – Sleep Health and Chronic Disease

