In April 2026, a new monoclonal antibody therapy demonstrated significant protection against Plasmodium falciparum malaria in children living in areas of intense, year-round transmission, offering a potential complement to existing prevention strategies like bed nets and seasonal chemoprevention. The therapy, which targets the circumsporozoite protein (CSP) on the surface of malaria sporozoites, works by blocking the parasite’s ability to invade liver cells—a critical early step in infection. This approach represents a shift from vaccine-induced immunity to direct passive immunization, providing immediate, high-titer antibody protection without relying on the recipient’s immune response.
How Monoclonal Antibodies Block Malaria Before It Takes Hold
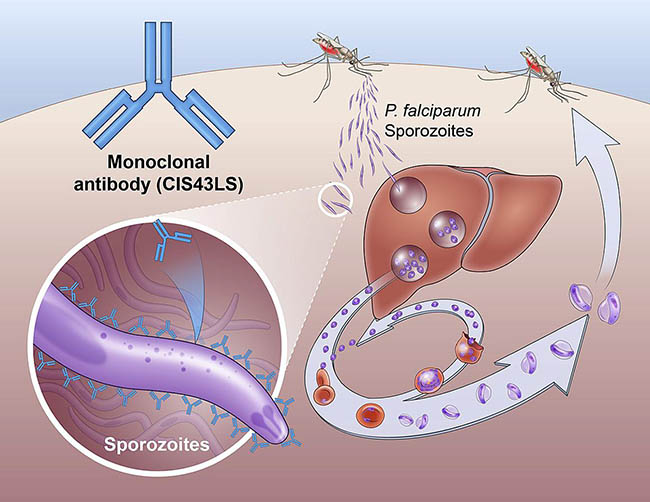
The monoclonal antibody candidate, CIS43LS, is a modified version of a naturally occurring human antibody identified from a volunteer who remained malaria-free despite exposure to infected mosquitoes. Through amino acid substitutions, scientists extended its half-life from weeks to months, enabling durable protection with infrequent dosing. In a Phase IIb trial conducted in Mali—a country with perennial malaria transmission and high inoculation rates—the antibody showed 75% efficacy against symptomatic P. Falciparum infection over a six-month malaria season. Unlike vaccines that require multiple doses and depend on robust immune responses, this approach delivers ready-made antibodies directly into the bloodstream, offering immediate defense. The mechanism is straightforward: the antibody binds to CSP, preventing the parasite from traversing the skin and infecting hepatocytes, thereby aborting the infection before blood-stage symptoms can develop.
In Plain English: The Clinical Takeaway
- This lab-made antibody can stop malaria parasites in the bloodstream before they reach the liver, where they would otherwise multiply and cause illness.
- In high-transmission areas like parts of Mali, a single injection provided protection for most children throughout the entire malaria season.
- Unlike vaccines, which take weeks to build immunity, this protection starts working within days and does not rely on the child’s own immune system to respond.
Real-World Impact in High-Burden Settings
Sub-Saharan Africa accounts for approximately 95% of global malaria cases and 96% of malaria deaths, with children under five bearing the greatest burden. In 2024, the World Health Organization estimated 249 million malaria cases worldwide, resulting in 608,000 deaths—over 80% of which occurred in just four African nations: Nigeria, the Democratic Republic of the Congo, Uganda, and Mali. Mali, where the CIS43LS trial was conducted, reports one of the highest entomological inoculation rates (EIR) globally, with some rural communities experiencing over 300 infectious mosquito bites per person per year during peak season. In such settings, even highly effective interventions like insecticide-treated nets (ITNs) and seasonal malaria chemoprevention (SMC) face limitations due to insecticide resistance, inconsistent adherence, and logistical challenges in reaching remote populations. The introduction of a long-acting monoclonal antibody could address these gaps, particularly for children who cannot access or tolerate seasonal chemoprevention drugs like sulfadoxine-pyrimethamine plus amodiaquine (SP-AQ) or dihydroartemisinin-piperaquine (DP).

“In areas where malaria transmission is intense and nearly constant, we need tools that operate independently of seasonal scheduling or patient adherence. A long-acting monoclonal antibody offers a precision prevention strategy that could protect the most vulnerable during peak transmission windows.”
Funding, Collaboration, and Regulatory Pathway
The clinical development of CIS43LS has been led by the Vaccine Research Center (VRC) at the National Institute of Allergy and Infectious Diseases (NIAID), part of the U.S. National Institutes of Health (NIH). The Phase IIb trial in Mali (NCT04244597) was conducted in collaboration with the University of Sciences, Techniques and Technologies of Bamako (USTTB) and supported by the NIH Intramural Research Program, with additional funding from the Bill & Melinda Gates Foundation through a grant to the Foundation for the National Institutes of Health (FNIH). No pharmaceutical company holds the patent; the technology is being advanced through public-private partnerships aimed at ensuring equitable access. As of early 2026, discussions are underway with the World Health Organization’s Prequalification Team and the European Medicines Agency (EMA) regarding potential regulatory pathways, though no formal submission has yet been made to the U.S. Food and Drug Administration (FDA) or EMA. Experts emphasize that any future deployment would require careful integration with existing malaria control programs to avoid fragmentation of services.
“The goal is not to replace bed nets or chemoprevention but to add a powerful layer of protection for those who fall through the cracks—children in remote villages, those with drug intolerances, or communities facing insecticide resistance. This represents about precision public health.”
Contraindications & When to Consult a Doctor
Monoclonal antibody therapies like CIS43LS are generally well-tolerated, with the most common adverse events being mild injection-site reactions (redness, swelling) and transient fever—occurring in less than 10% of recipients in clinical trials. Serious adverse events, including anaphylaxis or serum sickness, have not been observed in trials to date but remain theoretical risks with any exogenous protein therapy. There are no known contraindications based on age, sex, or comorbidities in the studied pediatric population (ages 5 to 12 months at enrollment), though safety data in infants under 5 months, pregnant individuals, or immunocompromised hosts are still limited. Parents should seek medical attention if a child develops persistent fever above 38.5°C, difficulty breathing, facial swelling, or widespread rash following administration. As with any new biologic, long-term monitoring for potential immune complex formation or delayed hypersensitivity is ongoing. Importantly, this therapy does not treat active malaria infection; children presenting with fever and suspected malaria should receive prompt diagnostic testing and artemisinin-based combination therapy (ACT) regardless of prophylaxis history.
| Parameter | Detail |
|---|---|
| Therapeutic Agent | CIS43LS (long-half-life monoclonal antibody targeting CSP) |
| Mechanism of Action | Blocks Plasmodium falciparum sporozoite invasion of hepatocytes |
| Trial Location | Mali (West Africa), perennial transmission setting |
| Target Population | Children aged 5 to 12 months at enrollment |
| Efficacy (6-month season) | 75% protection against symptomatic P. Falciparum infection |
| Dosing Regimen | Single intravenous infusion (5 mg/kg) |
| Half-Life Extension | Modified via LS mutation (half-life ~56 days) |
Future Outlook: Complement, Not Replace
While monoclonal antibody prophylaxis shows promise, it is not a standalone solution. Its high production cost, need for intravenous administration, and requirement for cold-chain storage pose significant barriers to widespread use in low-resource settings—challenges that also affect many biologics. Researchers are now exploring subcutaneous formulations and alternative delivery methods to improve feasibility. For now, experts agree that the greatest value lies in combining CIS43LS with existing tools: using it to protect high-risk subgroups during peak transmission, in conjunction with SMC, ITNs, and, where available, the RTS,S/AS01 or R21/Matrix-M malaria vaccines. The WHO’s 2023 recommendation for broader use of malaria vaccines in endemic regions underscores a shift toward layered, evidence-based prevention. As Dr. Djimdé noted, “No single tool will end malaria—but together, they can bring us closer than ever before.”
References
- Niang M, et al. CIS43LS monoclonal antibody prevents malaria infection in Mali. NEJM Evid. 2022;1(9).
- Wang C, et al. Potent human monoclonal antibodies protect mice against Plasmodium falciparum. Nature. 2021;593:130–135.
- World Health Organization. World Malaria Report 2024. Geneva: WHO; 2024.
- National Institutes of Health. National Institute of Allergy and Infectious Diseases (NIAID).
- Bill & Melinda Gates Foundation. Malaria Strategy Overview.

