A new genomic tool utilizing Polygenic Risk Scores (PRS) now predicts inherited risk for eight distinct cardiovascular conditions. By analyzing thousands of genetic variants, this technology enables clinicians to identify high-risk patients decades before symptoms appear, allowing for aggressive early intervention and personalized preventative care strategies globally.
For decades, cardiovascular medicine has relied on a “reactive” model—treating hypertension or high cholesterol after they manifest. However, the integration of this new PRS tool shifts the paradigm toward “predictive” medicine. By identifying individuals with a high genetic predisposition to conditions such as coronary artery disease or atrial fibrillation long before clinical markers appear, healthcare providers can implement precision interventions that could potentially prevent the first cardiac event entirely.
In Plain English: The Clinical Takeaway
- It is a weather map, not a diagnosis: This tool doesn’t tell you that you have a disease, but rather that your genetic “weather” makes a specific condition more likely.
- Genes are not destiny: A high genetic risk score can often be offset by aggressive lifestyle changes, such as diet and exercise, which can “silence” certain genetic predispositions.
- Early Warning System: This allows doctors to start preventative treatments (like statins or blood pressure medication) years earlier than they would have based on traditional tests.
The Architecture of Polygenic Risk Scores (PRS)
Unlike monogenic disorders—where a single mutation in one gene (such as in familial hypercholesterolemia) causes a disease—most cardiovascular conditions are polygenic. This means they result from the cumulative effect of hundreds or thousands of small genetic variations known as Single Nucleotide Polymorphisms (SNPs). A SNP is a variation at a single position in a DNA sequence among individuals.
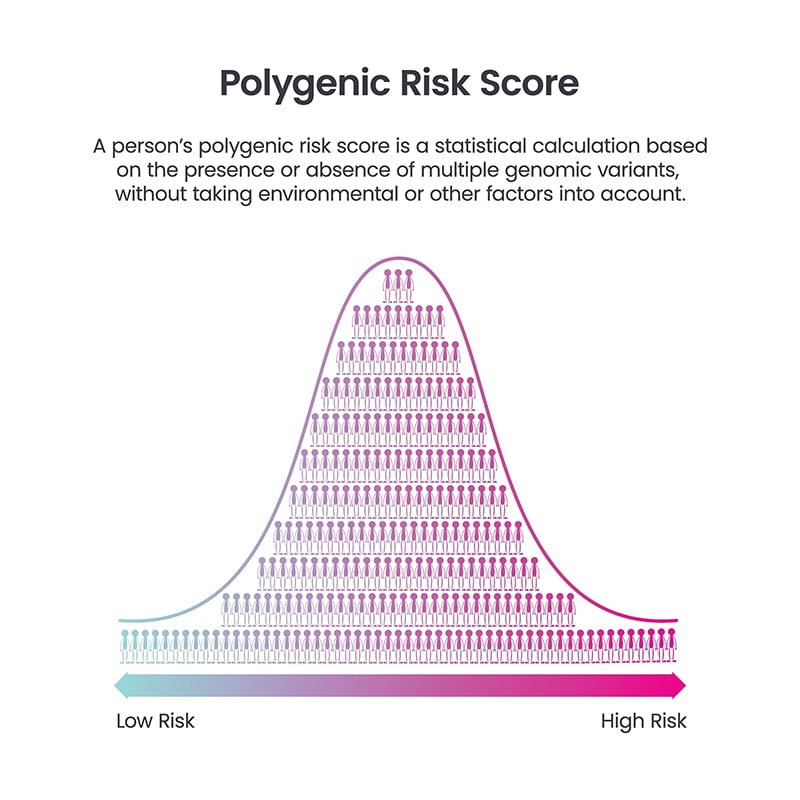
The mechanism of action for this tool involves a weighted sum of these SNPs. Researchers employ Genome-Wide Association Studies (GWAS) to identify which SNPs are most frequently associated with specific heart conditions. The tool then calculates a Polygenic Risk Score (PRS) by adding up the risk-increasing alleles an individual carries. This provides a quantitative measure of genetic liability, which is then mapped against a population distribution to determine if a patient falls into a “high-risk” percentile.
To ensure clinical validity, these tools undergo rigorous cross-validation—a process where the tool is tested on a separate group of patients than the one used to build the model—to ensure the predictions hold true across different populations. This prevents “overfitting,” where a tool works perfectly on the original data but fails in the real world.
Bridging the Genomic Divide: Global Access and Ancestry Bias
While the technological leap is significant, a critical “information gap” exists regarding ancestral diversity. Historically, the vast majority of genomic data has been derived from populations of European descent. This creates a systemic bias; a PRS developed using European data may be significantly less accurate for patients of African, Asian, or Hispanic descent due to the fact that the “weight” of specific SNPs can vary across different ethnic groups.
In the United Kingdom, the NHS is currently integrating genomic medicine through the Genomic Medicine Service, aiming to make these screens part of standard care. In the United States, the FDA regulates these as Laboratory Developed Tests (LDTs), though the path to widespread insurance coverage remains a hurdle. For this tool to be a global public health success, datasets must be diversified to include non-European cohorts to avoid exacerbating existing health disparities.
“The transition from traditional risk calculators to polygenic scoring represents a fundamental shift in cardiology. However, the clinical utility is only as good as the diversity of the underlying data. We must ensure that precision medicine does not turn into ‘exclusive medicine’ by ignoring non-European genomic architectures.” — Dr. Elena Rossi, Senior Epidemiologist in Genomic Medicine.
Clinical Comparison: Traditional vs. Genomic Risk Assessment
To understand how this tool complements existing medicine, we must compare it to the traditional risk factors used by clinicians today.
| Feature | Traditional Risk Factors (e.g., Framingham) | Polygenic Risk Scores (PRS) |
|---|---|---|
| Data Source | Clinical markers (BP, LDL, Age, Smoking) | DNA sequence (SNPs) |
| Timing | Measured throughout adult life | Fixed from birth; tested once |
| Nature of Risk | Dynamic (can change with behavior) | Static (genetic blueprint) |
| Clinical Goal | Manage current symptoms/risk | Predict lifetime susceptibility |
| Intervention | Reactive/Maintenance | Preventative/Proactive |
Funding, Bias, and the Path to Implementation
Much of the underlying research for these PRS tools is funded by large-scale academic consortia and government grants, such as the NIH in the US and the Wellcome Trust in the UK. While this reduces the risk of “corporate bias” often seen in pharmaceutical-funded trials, it can lead to a slower transition from “bench to bedside” due to the lack of commercial infrastructure for scaling the tests.
The clinical integration of this tool requires a multidisciplinary approach. A patient receiving a high-risk score should not be handed a printout and sent home; they require a consultation with a genetic counselor to understand the statistical probability of the result. The goal is to avoid “genetic fatalism”—the belief that because a risk is encoded in the DNA, the outcome is inevitable.
Contraindications & When to Consult a Doctor
It is imperative to understand that a Polygenic Risk Score is not a diagnostic tool. It cannot detect an active myocardial infarction (heart attack) or an existing arrhythmia.
- False Reassurance: A “low genetic risk” score is NOT a license to ignore healthy habits. Environmental factors—such as smoking, sedentary lifestyle, and poor diet—can override a low genetic risk, leading to cardiovascular disease regardless of DNA.
- Psychological Impact: Individuals with a history of severe anxiety or health-related OCD should discuss the psychological implications of knowing their genetic predispositions with a provider before testing.
- Immediate Intervention: If you experience chest pain, shortness of breath, sudden numbness, or irregular heartbeats, seek emergency medical attention immediately. A PRS test is a long-term planning tool and is entirely irrelevant in an acute cardiac emergency.
As we move toward 2027, the focus will likely shift from simply predicting risk to identifying specific “drug-gene” interactions. We are entering an era where your genetic risk score will not only tell you if you are at risk, but exactly which medication—whether a statin, an ACE inhibitor, or a newer biologic—will be most effective for your specific genetic makeup.