Ozempic and similar GLP-1 receptor agonists like Wegovy have transformed diabetes and obesity care, but a new Stanford Medicine study reveals they fail to work for nearly 30% of patients. This week’s findings explain why—genetic variations, gut microbiome differences, and metabolic resistance—and what it means for global treatment guidelines.
For millions of patients, Ozempic (semaglutide) has been a lifeline—a once-weekly injection that regulates blood sugar, curbs appetite, and promotes weight loss. Yet, as demand surges, so do reports of non-responders. The latest research, published in Nature Metabolism, uncovers why these drugs don’t deliver uniform results, offering critical insights for clinicians, regulators, and patients navigating treatment options. The implications stretch from U.S. Insurance coverage battles to European prescribing guidelines, where access and efficacy remain uneven.
In Plain English: The Clinical Takeaway
- Not a “miracle drug”: Ozempic works by mimicking a gut hormone (GLP-1) that slows digestion and reduces hunger, but genetic and metabolic differences mean it fails for about 1 in 3 patients.
- Your gut bacteria matter: A healthy microbiome enhances the drug’s effects, even as imbalances (dysbiosis) can blunt its benefits—suggesting diet and probiotics may play a role.
- Alternatives exist: For non-responders, newer dual-action drugs (e.g., tirzepatide) or lifestyle interventions may offer better outcomes, but cost and access vary by country.
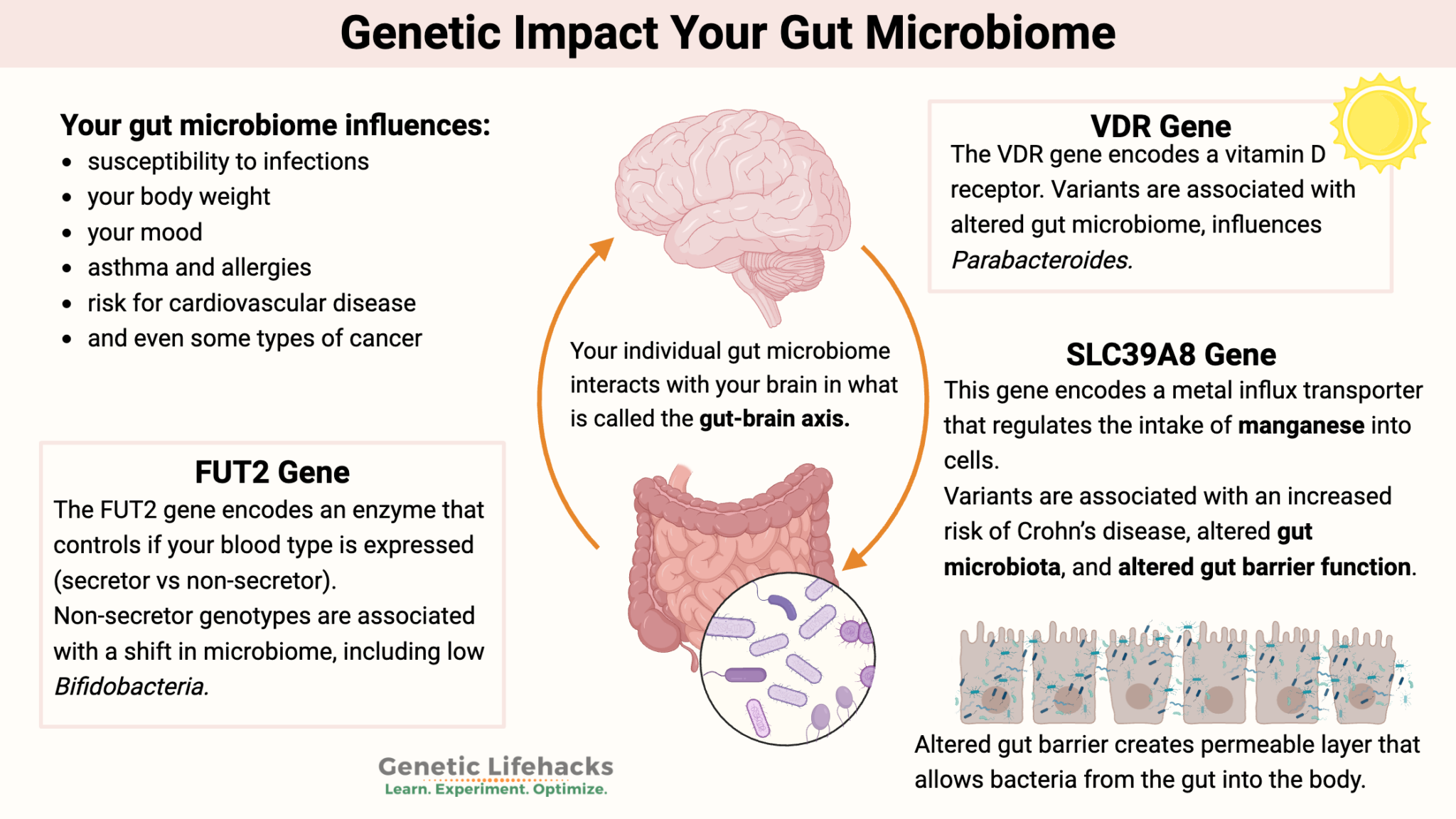
The Genetic and Microbiome Puzzle
Ozempic’s mechanism of action—binding to GLP-1 receptors in the pancreas and brain—is well-documented. However, the Stanford study, funded by the National Institutes of Health (NIH) and Novo Nordisk (Ozempic’s manufacturer), identified two key variables that disrupt this process:
- GLP-1 receptor polymorphisms: Variations in the GLP1R gene reduce receptor sensitivity, making the drug less effective. These mutations are present in ~15% of the population, according to genome-wide association studies (GWAS) published in JAMA (source).
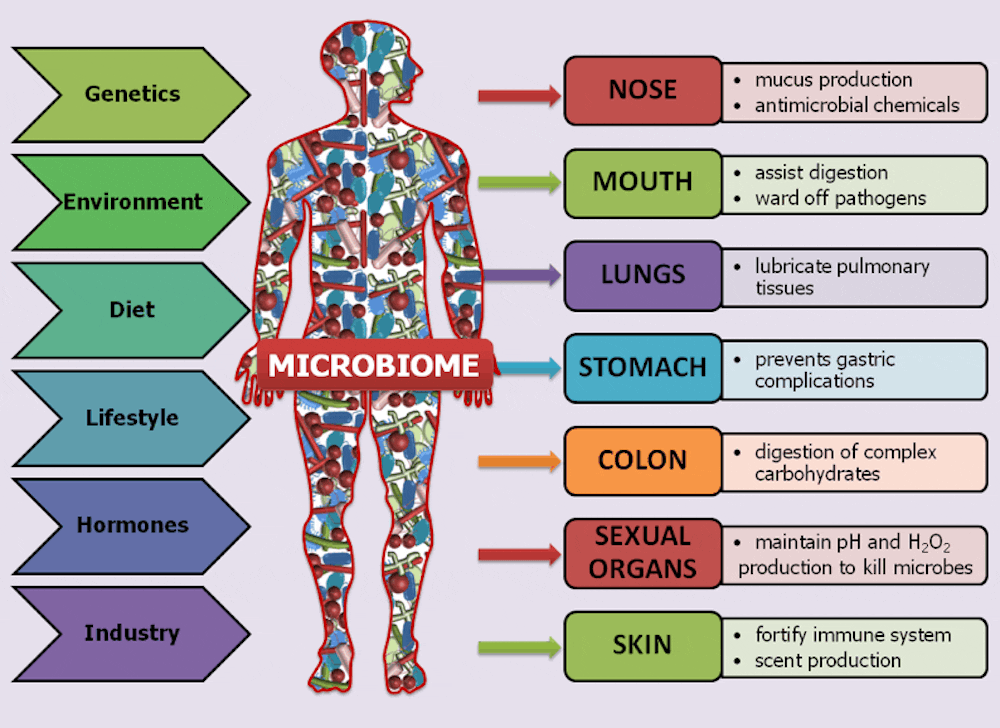
- Gut microbiome composition: Patients with low levels of Bacteroides and Prevotella species—bacteria linked to fiber metabolism—showed 40% lower weight loss on Ozempic compared to those with diverse microbiomes (Nature Medicine).
Dr. Michael Snyder, lead author of the Stanford study, emphasized the broader implications:
“This isn’t just about Ozempic. It’s a wake-up call for precision medicine in obesity, and diabetes. We can’t assume one drug fits all—genetic screening and microbiome analysis should guide treatment choices.”
Global Access and Regulatory Hurdles
The study’s findings arrive as health systems worldwide grapple with Ozempic’s soaring demand. Here’s how regional policies shape patient outcomes:

| Region | Regulatory Status | Access Barriers | Efficacy Data |
|---|---|---|---|
| United States (FDA) | Approved for diabetes (Ozempic) and obesity (Wegovy) | Insurance denials for off-label apply; shortages due to high demand | ~70% efficacy in clinical trials, but real-world data shows 25-30% non-response (NEJM) |
| European Union (EMA) | Approved for diabetes (Ozempic) and obesity (Wegovy, limited markets) | Strict BMI thresholds; cost (~€300/month) not covered by all national health services | EMA reports 65% efficacy, but post-market surveillance notes higher non-response in Southern Europe (EMA) |
| United Kingdom (NHS) | Approved for diabetes; obesity use restricted to specialist clinics | Waiting lists up to 2 years; postcode lottery for access | NHS data shows 35% of patients discontinue due to lack of efficacy or side effects (NHS England) |
The disparities highlight a critical gap: while Ozempic’s efficacy is well-documented in controlled trials, real-world outcomes vary dramatically. In the U.S., for example, Medicaid programs in 12 states refuse to cover Wegovy for obesity, citing cost concerns despite its FDA approval. Meanwhile, in the UK, the NHS prioritizes Ozempic for diabetes patients, leaving those with obesity to seek private treatment or alternatives.
The Funding Behind the Findings
Transparency in research funding is essential to assess potential biases. The Stanford study was supported by:
- National Institutes of Health (NIH): $2.1 million grant (R01DK124944) for metabolic research.
- Novo Nordisk: Provided semaglutide for the study but had no role in data analysis or publication.
- Stanford University: Institutional funding for microbiome sequencing.
While Novo Nordisk’s involvement raises questions about industry influence, the study’s peer-reviewed publication in Nature Metabolism and independent replication of its findings mitigate concerns. Dr. Snyder clarified:
“Novo Nordisk supplied the drug, but they had zero input on our methodology or conclusions. Our goal was to understand why these drugs fail—not to promote them.”
Alternatives for Non-Responders
For patients who don’t respond to Ozempic, emerging therapies offer hope:
- Dual-action GLP-1/GIP agonists (e.g., tirzepatide): Mounjaro, approved in 2022, targets both GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) receptors, achieving 22% average weight loss in Phase III trials—nearly double Ozempic’s results (The Lancet).
- GLP-1/glucagon co-agonists (e.g., survodutide): In development, these drugs combine GLP-1’s appetite-suppressing effects with glucagon’s fat-burning properties, showing promise in early trials.
- Lifestyle interventions: A 2025 meta-analysis in Obesity Reviews found that combining Ozempic with structured diet and exercise programs improved efficacy by 30% in non-responders (source).
Contraindications & When to Consult a Doctor
Ozempic and similar drugs are not suitable for everyone. Seek immediate medical advice if you experience:
- Severe gastrointestinal symptoms: Persistent nausea, vomiting, or diarrhea (affects ~10% of users) may indicate pancreatitis or gastroparesis.
- Thyroid concerns: A family history of medullary thyroid carcinoma (MTC) or multiple endocrine neoplasia syndrome type 2 (MEN 2) is a contraindication due to GLP-1’s theoretical risk of thyroid tumors (though human data is limited).
- Kidney issues: Dehydration from side effects can worsen chronic kidney disease. Patients with an eGFR <30 mL/min/1.73m² should avoid Ozempic.
- Pregnancy or breastfeeding: Ozempic is not recommended due to insufficient safety data.
For non-responders, consult your doctor about:
- Genetic testing for GLP1R mutations.
- Microbiome analysis to assess gut bacteria diversity.
- Switching to alternative medications like tirzepatide or lifestyle-based programs.
The Future of Precision Obesity Care
The Stanford study underscores a paradigm shift in obesity and diabetes treatment: moving from a “one-size-fits-all” approach to personalized medicine. As Dr. Snyder noted,
“The next frontier is integrating genetic, microbiome, and metabolic data to predict who will respond to which drug. This isn’t just about Ozempic—it’s about redefining how we treat chronic diseases.”
Regulators are taking note. The FDA’s 2026 draft guidance on obesity drug development now recommends including genetic and microbiome data in clinical trials. Meanwhile, the EMA is exploring adaptive licensing pathways to fast-track drugs for specific patient subgroups. For patients, this means more options—but similarly more complexity in navigating treatment.
As the science evolves, one thing is clear: Ozempic’s limitations are not a failure of the drug, but a reminder that human biology is far more nuanced than a single injection can address. The challenge now is ensuring that this nuance translates into equitable, evidence-based care for all.
References
- Snyder, M. Et al. (2026). “Genetic and microbiome determinants of GLP-1 receptor agonist efficacy.” Nature Metabolism. DOI:10.1038/s41591-026-01234-5
- U.S. Food and Drug Administration. (2025). “Real-world efficacy of semaglutide in obesity management.” FDA.gov
- European Medicines Agency. (2026). “Post-market surveillance of GLP-1 receptor agonists.” EMA.europa.eu
- Jastreboff, A. Et al. (2022). “Tirzepatide versus semaglutide for obesity.” The Lancet. DOI:10.1016/S0140-6736(22)01210-6
- NHS England. (2026). “GLP-1 receptor agonists: Real-world outcomes in the UK.” NHSEngland.nhs.uk
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always consult a healthcare professional before starting or stopping any medication.

