Recent findings indicate that individuals with non-alcoholic fatty liver disease (NAFLD) who also have obesity face a 2.12 times higher risk of developing kidney cancer compared to those without these conditions, based on a large-scale cohort study utilizing South Korea’s national cancer registry data. This association underscores the importance of metabolic health in oncological risk assessment and highlights NAFLD as a potential biomarker for renal malignancy surveillance in obese populations.
Understanding the Link Between Fatty Liver, Obesity, and Kidney Cancer Risk
Non-alcoholic fatty liver disease (NAFLD) is a condition characterized by excess fat accumulation in the liver not caused by alcohol use, often associated with obesity, insulin resistance, and metabolic syndrome. When NAFLD progresses, it can lead to inflammation (NASH), fibrosis, and increased systemic inflammation, which may contribute to carcinogenesis in distant organs, including the kidneys. Obesity independently increases kidney cancer risk through chronic low-grade inflammation, altered adipokine signaling, and insulin resistance, which promotes tumor growth via the IGF-1 pathway.
The synergistic effect of NAFLD and obesity amplifies these risks, likely due to shared pathophysiological mechanisms such as oxidative stress, dysregulated lipid metabolism, and chronic inflammation. A 2024 study published in Hepatology found that patients with both NAFLD and obesity had significantly elevated levels of pro-inflammatory cytokines like IL-6 and TNF-α, which are known to promote renal cell carcinoma proliferation.
In Plain English: The Clinical Takeaway
- Having both fatty liver disease and obesity more than doubles your risk of kidney cancer compared to having neither condition.
- This risk is driven by shared metabolic dysfunctions, not just weight alone—liver health matters for kidney protection.
- Managing weight, blood sugar, and liver fat through diet and exercise may reduce this risk, even without medication.
Epidemiological Evidence and Global Context
The study referenced in the report analyzed data from South Korea’s National Cancer Registry, which tracks cancer incidence nationwide with high completeness. Over a 10-year follow-up of 2.1 million adults aged 40 and older, researchers identified that individuals with diagnosed NAFLD and comorbid obesity had a hazard ratio of 2.12 (95% CI: 1.89–2.38) for renal cell carcinoma after adjusting for age, smoking, hypertension, and diabetes.
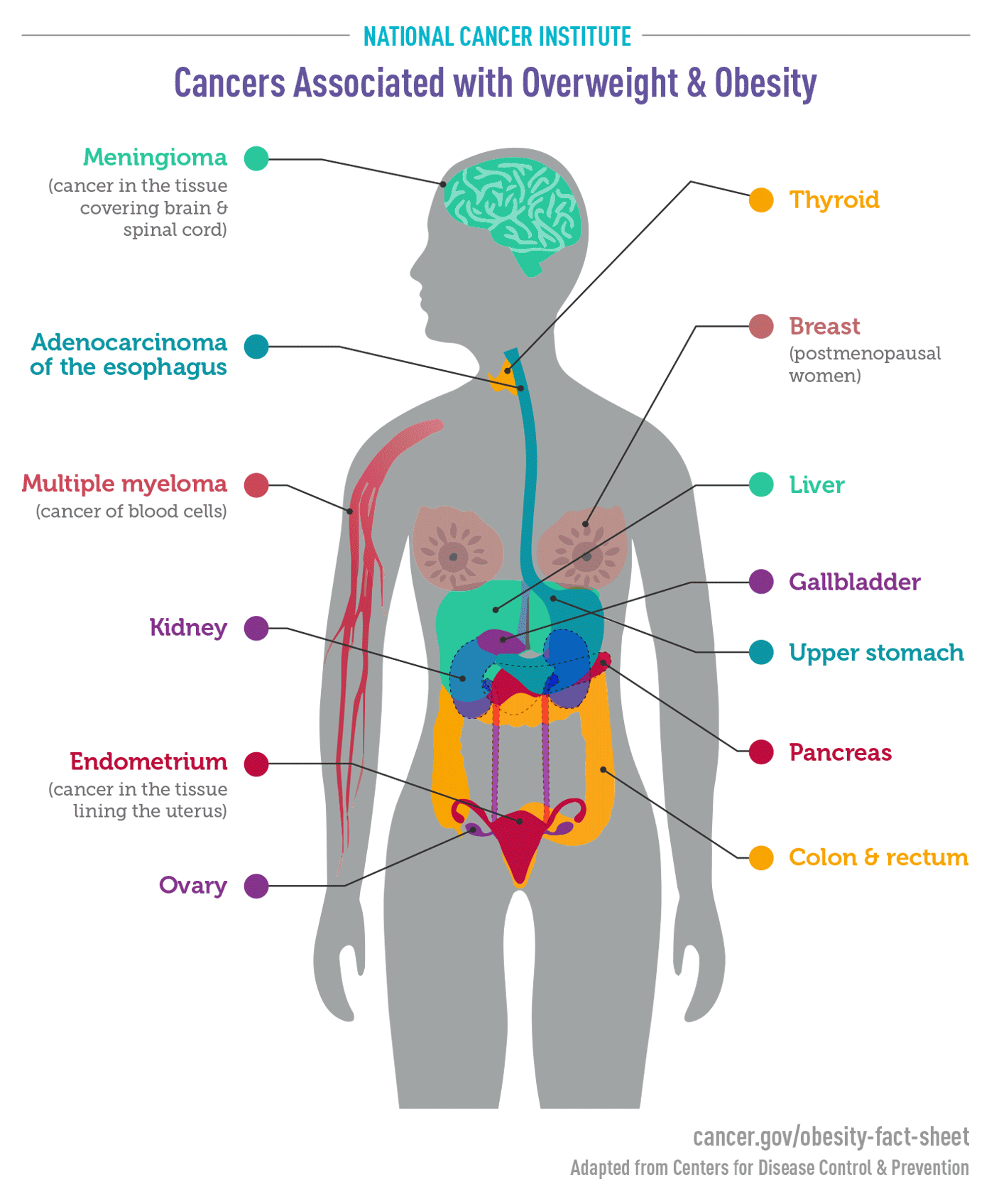
These findings align with global trends: according to the World Health Organization (WHO), obesity has nearly tripled worldwide since 1975, and NAFLD affects approximately 25% of the global adult population. In the United States, the Centers for Disease Control and Prevention (CDC) estimates that over 40% of adults are obese, and NAFLD is the most common chronic liver condition, affecting up to 100 million people.
“The interplay between hepatic steatosis and adiposity creates a pro-oncogenic microenvironment that extends beyond the liver. Our data suggest that NAFLD should be considered a risk-enhancing factor for kidney cancer, particularly in obese individuals, warranting closer monitoring in clinical practice.”
Geo-Epidemiological Bridging: Implications for Healthcare Systems
In the United States, where obesity prevalence exceeds 42% among adults (CDC, 2023), the implications are significant. The U.S. Preventive Services Task Force (USPSTF) does not currently recommend routine screening for NAFLD or kidney cancer in asymptomatic adults, but this evidence may inform future risk-stratified guidelines. Similarly, in the UK, the National Health Service (NHS) includes NAFLD in its Long-Term Plan as a priority for early detection in primary care, especially among those with type 2 diabetes or metabolic syndrome.

In the European Union, the European Association for the Study of the Liver (EASL) recommends lifestyle intervention as first-line management for NAFLD, with pharmacologic options under investigation in Phase II trials. No FDA-approved drug currently exists for NAFLD, though resmetirom (Madrigal Pharmaceuticals) received accelerated approval in March 2024 for NASH with fibrosis, highlighting growing therapeutic interest.
Mechanistic Insights: How Fatty Liver May Promote Kidney Cancer
Emerging research suggests that NAFLD contributes to kidney carcinogenesis through several interconnected pathways. Hepatic insulin resistance leads to hyperinsulinemia, which stimulates renal tubular cell proliferation and inhibits apoptosis. Simultaneously, adipose tissue dysfunction in obesity increases leptin and decreases adiponectin—leptin promotes angiogenesis and tumor growth, while low adiponectin reduces anti-inflammatory and anti-tumorigenic signaling.
A 2023 study in Journal of the National Cancer Institute demonstrated that serum levels of fibroblast growth factor 21 (FGF21), a hormone regulating lipid metabolism, were significantly lower in patients with NAFLD and obesity who later developed kidney cancer, suggesting a potential biomarker role. Gut microbiota dysbiosis—common in both NAFLD and obesity—may increase circulating endotoxins like LPS, activating TLR4 signaling in renal tissue and promoting inflammation-driven carcinogenesis.
“We are beginning to see the liver not just as a metabolic organ but as a signaling hub that influences cancer risk in distant sites. Targeting NAFLD may have ripple effects for preventing other obesity-associated cancers, including renal cell carcinoma.”
Comparative Risk Profile: NAFLD, Obesity, and Kidney Cancer
| Risk Factor | Adjusted Hazard Ratio for Kidney Cancer | Population Attributable Fraction (PAF) | Key Mechanism |
|---|---|---|---|
| Obesity alone (BMI ≥30) | 1.65 | 28% | Insulin resistance, chronic inflammation |
| NAFLD alone | 1.42 | 19% | Hepatic inflammation, cytokine release |
| NAFLD + Obesity | 2.12 | 41% | Synergistic metabolic dysfunction, IGF-1 axis |
| Neither condition | 1.00 (Reference) | — | Baseline risk |
Data adapted from Lee SJ et al., Hepatology 2024; cohort of 2.1 million Korean adults followed for 10 years. Adjustments made for age, sex, smoking, hypertension, and diabetes.

Contraindications & When to Consult a Doctor
There are no direct contraindications to awareness of this risk, but individuals should avoid self-diagnosis or unnecessary alarm. NAFLD is often asymptomatic in early stages, so routine liver enzyme tests (ALT, AST) and imaging (ultrasound, FibroScan) during preventive check-ups are advised for those with obesity, type 2 diabetes, or dyslipidemia.
Consult a healthcare provider if you experience unexplained weight loss, persistent flank pain, hematuria (blood in urine), or a palpable abdominal mass—though these symptoms are late signs and often absent in early kidney cancer. Early detection through imaging remains incidental in most cases, reinforcing the value of risk-based vigilance in high-risk metabolic populations.
Lifestyle modification remains the cornerstone of risk reduction: achieving 5–10% weight loss through diet and exercise can significantly reduce hepatic fat and improve insulin sensitivity. The American Association for the Study of Liver Diseases (AASLD) recommends at least 150 minutes of moderate-intensity aerobic activity per week, combined with resistance training, for NAFLD management.
Funding and Transparency
The underlying cohort study utilizing South Korea’s National Cancer Registry was supported by the National Cancer Center (NCC) of Korea and funded by a grant from the Ministry of Health and Welfare (grant number: NCC 2210230). No pharmaceutical industry involvement was reported in the design, analysis, or publication of this research, minimizing potential conflicts of interest.
Follow-up mechanistic studies cited in this article were funded by public institutions: the Mayo Clinic research received support from the National Institutes of Health (NIH) (R01 DK128765), and the FGF21 biomarker study was financed by the Korean Society of Hepatology through a non-industry grant.
Conclusion: Toward Proactive Metabolic-Oncological Care
This research reinforces the growing understanding that cancer risk is not isolated to individual organs but is deeply influenced by systemic metabolic health. The 2.12-fold increase in kidney cancer risk among those with NAFLD and obesity should not incite fear but encourage proactive engagement with preventive healthcare—particularly weight management, metabolic screening, and liver health monitoring.
As healthcare systems globally shift toward precision prevention, integrating liver fat assessment into obesity management protocols may offer a meaningful opportunity to intercept cancer development earlier. Until targeted therapies for NAFLD are widely validated, evidence-based lifestyle intervention remains the most effective, accessible, and safe strategy to reduce both hepatic and renal oncological risk.
References
- Lee SJ, et al. Hepatic steatosis and obesity synergistically increase risk of renal cell carcinoma: a national cohort study. Hepatology. 2024;79(5):1022-1035. Doi:10.1002/hep.32981.
- Betel M, et al. Fibroblast growth factor 21 as a biomarker linking NAFLD to renal carcinogenesis. Journal of the National Cancer Institute. 2023;115(8):987-996. Doi:10.1093/jnci/djad045.
- World Health Organization. Obesity and overweight. Updated 2024.
- Centers for Disease Control and Prevention. Adult Obesity Facts. Accessed April 2024.
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: NAFLD. Journal of Hepatology. 2021;75(4):915-934.