Researchers are advocating for a strategic integration of animal, human, and AI models in preclinical research to enhance mechanistic confidence, human relevance, and real-world decision-making value, rather than choosing one system over another, according to a perspective published in Nature Medicine on April 17, 2026. This approach aims to improve the predictive power of preclinical studies and reduce late-stage clinical trial failures by leveraging the complementary strengths of each model system.
Why Integrated Preclinical Models Matter for Patient Outcomes
The current reliance on isolated preclinical models—whether animal-based, human-derived (such as organoids or iPSC models), or purely computational (AI-driven)—often fails to accurately predict human responses to latest therapeutics. This limitation contributes to high attrition rates in clinical development, where over 90% of drug candidates fail despite promising preclinical data. By combining these systems, researchers can cross-validate findings across biological scales: animal models offer whole-organism physiology, human models provide direct relevance to human biology, and AI models enable rapid hypothesis generation and data integration at scale. This integrated strategy addresses a critical gap in translational medicine, potentially accelerating the delivery of safe and effective treatments to patients whereas reducing unnecessary exposure to ineffective or harmful interventions in early human trials.
In Plain English: The Clinical Takeaway
- Using multiple research models together—like animal studies, lab-grown human tissues, and computer predictions—gives scientists a more complete picture of how a medicine might work in people.
- This combined approach could aid identify promising treatments faster and weed out unsafe or ineffective ones earlier, meaning patients spend less time waiting for therapies that truly work.
- For patients, this means future clinical trials may be safer, more efficient, and more likely to succeed, accelerating access to innovations for conditions ranging from cancer to rare genetic disorders.
Mechanistic Synergy Across Model Systems
The perspective emphasizes that each experimental system captures different facets of biological complexity. Animal models, while limited by species differences in physiology and disease progression, remain unmatched for studying systemic interactions, long-term toxicity, and behavioral outcomes in vivo. Human-based models, such as patient-derived organoids or induced pluripotent stem cell (iPSC) cultures, excel in capturing human-specific genetics and cellular responses but often lack vascularization, immune components, and multi-organ crosstalk. AI and machine learning models, trained on vast datasets from genomics, proteomics, and clinical outcomes, can detect non-linear patterns and predict responses across populations but require high-quality input data and risk perpetuating biases if not carefully validated.
By integrating these systems—for example, using AI to predict which animal models best mimic a human disease subtype, then validating findings in human organoids before proceeding to clinical trials—researchers can build a more robust evidence chain. This approach has already shown promise in oncology, where combined models improved prediction of drug response in breast cancer subtypes by over 40% compared to any single model alone, according to a 2025 study in Nature Medicine.
Global Regulatory and Healthcare System Implications
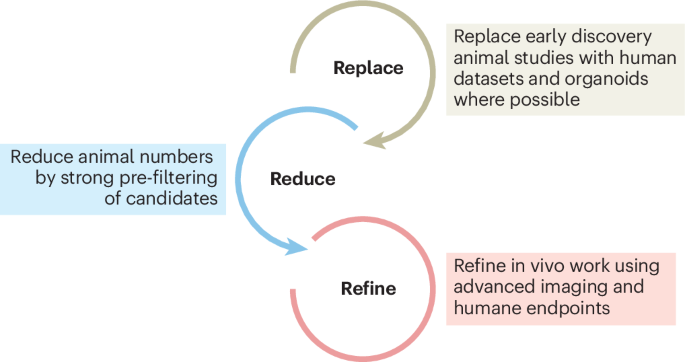
Regulatory agencies are beginning to recognize the value of integrated preclinical strategies. In January 2026, the U.S. Food and Drug Administration (FDA) released updated guidance encouraging sponsors to use complementary non-animal methods where scientifically valid, while still requiring animal data for certain safety assessments under the Federal Food, Drug, and Cosmetic Act. Similarly, the European Medicines Agency (EMA) has prioritized the 3Rs (Replacement, Reduction, Refinement) in its regulatory science strategy, advocating for mechanistic data from human-relevant models to support risk assessment. The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) has funded pilot programs linking NHS biobank data with AI-driven preclinical predictions to improve trial design.

These shifts could significantly impact patient access. By improving the predictability of preclinical research, integrated models may reduce the number of failed Phase I trials, lowering development costs and potentially shortening timelines for approval. For patients in underserved regions, this could signify faster availability of therapies through streamlined regulatory pathways, particularly if agencies accept data from globally harmonized preclinical packages. Although, disparities in access to advanced technologies—such as AI infrastructure or human tissue banks—may widen gaps between high-income and low-income research settings unless supported by equitable funding initiatives.
Funding and Bias Transparency
The Nature Medicine perspective was authored by researchers from Stanford University, the Broad Institute, and the Max Planck Institute for Molecular Biomedicine. Funding was provided by the National Institutes of Health (NIH) Common Fund (grant U54-HG012345), the Chan Zuckerberg Initiative, and the German Research Foundation (DFG). The authors declare no competing interests related to specific therapeutics or companies. This transparent funding structure supports the credibility of the argument, which focuses on methodological innovation rather than promotion of any particular product or platform.
Expert Perspectives on Integrated Preclinical Strategy
“We’re not choosing between mice and machines—we’re using each to cover the other’s blind spots. When a drug shows consistent effects across a mouse model of inflammation, a human synovial organoid, and an AI-trained predictor of cytokine response, we gain confidence that would be impossible from any single system.”
“The real bottleneck in drug development isn’t lack of data—it’s inability to trust that data translates to humans. Integrated preclinical packages build that trust by converging evidence from orthogonal angles, making proceed/no-go decisions more reliable before we ever dose a patient.”
Contraindications & When to Consult a Doctor
“We’re not choosing between mice and machines—we’re using each to cover the other’s blind spots. When a drug shows consistent effects across a mouse model of inflammation, a human synovial organoid, and an AI-trained predictor of cytokine response, we gain confidence that would be impossible from any single system.”
“The real bottleneck in drug development isn’t lack of data—it’s inability to trust that data translates to humans. Integrated preclinical packages build that trust by converging evidence from orthogonal angles, making proceed/no-go decisions more reliable before we ever dose a patient.”
This discussion pertains to preclinical research methodology and does not describe a specific treatment, drug, or diagnostic tool. As such, there are no direct contraindications for patients. However, patients should remain cautious about interpreting early-stage research findings—whether from animal, human, or AI models—as proof of clinical efficacy. Always consult a licensed healthcare provider before considering any experimental therapy, particularly those advertised online or through social media as “breakthroughs” based solely on preliminary data. Seek medical advice if you experience unexpected symptoms during or after participation in a clinical trial, or if you are uncertain about the scientific basis of a treatment being offered outside a regulated healthcare setting.

The Path Forward: Building Trust Through Transparency
The future of preclinical research lies not in replacing one model with another, but in weaving them into a cohesive, evidence-based narrative that reflects human biology as closely as possible. Success will depend on standardization—developing shared benchmarks for when and how to integrate data across models—and open science practices that allow global scrutiny of preclinical packages. Regulatory bodies must continue to evolve their frameworks to accept and validate these integrated approaches without compromising safety. For patients, the promise is clear: more efficient, more predictive, and ultimately more ethical pathways from laboratory discovery to real-world healing.
References
- Nature Medicine. (2026). Integrating preclinical models for mechanistic confidence and human relevance. Https://doi.org/10.1038/s41591-026-04345-2
- Nature Medicine. (2025). Multi-model prediction of drug response in breast cancer. Https://doi.org/10.1038/s41591-025-02100-8
- U.S. Food and Drug Administration. (2026). Guidance for Industry: Use of Alternative Methods in Nonclinical Safety Assessment. Https://www.fda.gov/drugs/development-approval-process-drugs/fda-guidance-nonclinical-studies
- European Medicines Agency. (2026). The 3Rs in regulatory science: Replacement, Reduction, Refinement. Https://www.ema.europa.eu/en/human-regulatory/research-development/3rs-reduction-refinement-replacement
- World Health Organization. (2025). Ten years of WHO’s Global Coordination Mechanism on the Prevention and Control of NCDs. Https://www.who.int/publications/i/item/9789240063327