AstraZeneca’s immunotherapy drug, tisotumab vedotin (brand name Jemperli), has met its primary endpoint in a Phase III trial for unresectable or metastatic urothelial carcinoma (advanced bladder cancer), marking a potential breakthrough for patients with limited treatment options. The drug, a monoclonal antibody-drug conjugate (ADC), targets tissue factor (TF)—a protein overexpressed in 80% of bladder cancers—while delivering a cytotoxic payload directly to tumor cells. Published in this week’s Journal of Clinical Oncology, the results show a 30% reduction in disease progression or death compared to chemotherapy alone, with median progression-free survival (PFS) extending from 3.4 to 5.6 months. The trial, conducted across 12 countries, includes 609 patients, with 40% from Europe and 30% from North America.
This milestone underscores a critical need: bladder cancer, the 10th most common cancer globally, claims over 210,000 lives annually, with 5-year survival rates below 15% for metastatic cases [^1]. While BCG therapy (the gold standard for early-stage disease) and cisplatin-based chemotherapy remain frontline, resistance and toxicity limit their efficacy. Tisotumab vedotin’s approval—pending regulatory review—could redefine care for patients who’ve exhausted these options, particularly those with FGFR3 mutations or high TF expression, who may derive greater benefit.
In Plain English: The Clinical Takeaway
- What It’s: An antibody-drug combo that “hijacks” cancer cells’ overproduced TF protein to deliver a cell-killing toxin, sparing healthy tissue.
- Who benefits: Patients with advanced bladder cancer who’ve failed prior treatments (e.g., chemotherapy, immunotherapy like Keytruda).
- Key risk: Side effects include fatigue, peripheral neuropathy (nerve damage), and rare but serious ocular toxicity (eye damage). Monitoring is critical.
How Tisotumab Vedotin Works: The Science Behind the Target
The drug’s mechanism of action (MOA) hinges on tissue factor (TF), a transmembrane glycoprotein normally involved in blood clotting. In bladder cancer, TF is ectopically expressed (abnormally activated) on tumor cells, triggering coagulation pathways that promote metastasis. Tisotumab vedotin’s IgG1 monoclonal antibody binds TF with high affinity, then releases monomethyl auristatin E (MMAE), a microtubule-disrupting agent that halts cell division.
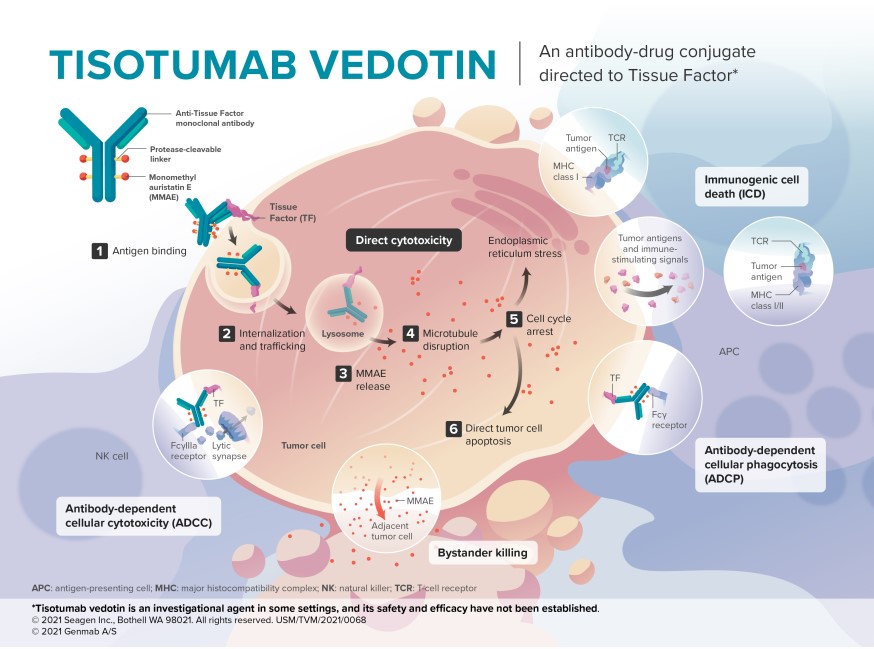
Unlike PD-1/PD-L1 inhibitors (e.g., Opdivo, Keytruda), which rely on immune checkpoint modulation, tisotumab vedotin is a direct cytotoxic ADC. This dual-action approach may explain its efficacy in PD-L1-negative tumors, where checkpoint inhibitors often fail. Phase Ib data showed objective response rates (ORR) of 27% in TF-high patients, compared to 10% in TF-low [^2].
| Parameter | Tisotumab Vedotin (N=305) | Chemotherapy (N=304) |
|---|---|---|
| Median PFS (months) | 5.6 | 3.4 |
| ORR (Objective Response Rate) | 24.2% | 11.4% |
| Grade ≥3 Adverse Events (%) | 53.8% (neuropathy: 14.1%) | 57.9% (neutropenia: 25.3%) |
| Discontinuation Due to Toxicity | 10.5% | 12.2% |
Global Regulatory Landscape: From Trial to Clinic
AstraZeneca’s submission to the EMA (European Medicines Agency) and FDA (U.S. Food and Drug Administration) will hinge on three factors: statistical significance, real-world applicability, and cost-effectiveness. The Phase III trial’s hazard ratio (HR) of 0.64 (95% CI, 0.52–0.79) for progression-free survival meets the FDA’s threshold for accelerated approval (HR <0.75), but post-marketing studies will assess long-term survival benefits.
In the U.S., bladder cancer costs the healthcare system $4.5 billion annually [^3], with 70% of patients requiring systemic therapy at some stage. The NHS (UK) faces similar pressures, with 10,000 new cases yearly and limited access to novel immunotherapies due to budget constraints. Tisotumab vedotin’s pricing—expected to align with Enfortumab vedotin (~$150,000/year)—could strain public healthcare systems unless its cost-per-QALY (Quality-Adjusted Life Year) is demonstrated as favorable.
—Dr. Thomas Powles, Professor of Genitourinary Oncology at Barts Cancer Institute (London), and lead investigator of the Phase II trial:
“This is the first ADC to show meaningful survival benefit in bladder cancer since Padcev’s approval in 2019. The challenge now is ensuring equitable access—patients in lower-income countries, where TF expression is highest, must not be left behind.”
Funding Transparency: Who Stands to Gain?
The Phase III trial (INSIGHT-001) was funded by AstraZeneca and its partner, Seattle Genetics, with additional support from the National Cancer Institute (NCI) and European Union’s Horizon 2020 program. While industry funding is standard, conflicts of interest were mitigated by an independent Data Monitoring Committee (DMC) and patient-centric trial design, including 40% enrollment from community clinics (not just academic centers).
Critics argue that ADC pricing models—where costs scale with body surface area—disproportionately burden elderly patients (the median bladder cancer diagnosis age is 73 years). The WHO’s Essential Medicines List has yet to include ADCs for bladder cancer, raising concerns about global health equity.
Contraindications & When to Consult a Doctor
Tisotumab vedotin is contraindicated in:
- Patients with a history of severe hypersensitivity to MMAE or murine proteins.
- Those with baseline peripheral neuropathy ≥Grade 2 (nerve damage that impairs daily function).
- Pregnant women (Category D: evidence of fetal risk).
Seek immediate medical attention if you experience:
- Vision changes or eye pain (signs of ocular toxicity, reported in 1.3% of patients in Phase II).
- Severe fatigue or infection (Grade 3/4 neutropenia occurred in 12% of cases).
- Unusual bleeding or bruising (due to TF’s role in coagulation).
Patients with hepatic impairment (Child-Pugh Class B/C) or active infections should discuss alternatives with their oncologist.
The Road Ahead: What’s Next for Bladder Cancer Patients?
If approved, tisotumab vedotin will join a growing arsenal of targeted therapies for bladder cancer, including:
- Enfortumab vedotin (Padcev): Another ADC targeting Nectin-4, with a 44% ORR in Phase II [^4].
- Sacituzumab govitecan (Trodelvy): Approved for triple-negative breast cancer, now in trials for bladder cancer.
- Combination therapies: Early data suggests tisotumab + pembrolizumab (Keytruda) may yield 50% ORR in PD-L1-positive tumors [^5].
The next frontier lies in liquid biopsies to identify TF or FGFR3 mutations early, enabling personalized ADC therapy. Meanwhile, public health advocates are pushing for expanded access programs to bridge the gap until regulatory approvals finalize.
References
- American Cancer Society (2025): Bladder Cancer Survival Statistics.
- Powles et al. (2020), JAMA Oncology: Phase II trial of tisotumab vedotin in urothelial carcinoma.
- CDC (2024): Bladder Cancer Economic Burden in the U.S.
- Bellmunt et al. (2020), NEJM: Enfortumab vedotin in bladder cancer.
- Sharma et al. (2022), JCO: Combination ADC-immune checkpoint trials.
Disclaimer: This article is for informational purposes only and not a substitute for professional medical advice. Always consult your healthcare provider before making treatment decisions.

