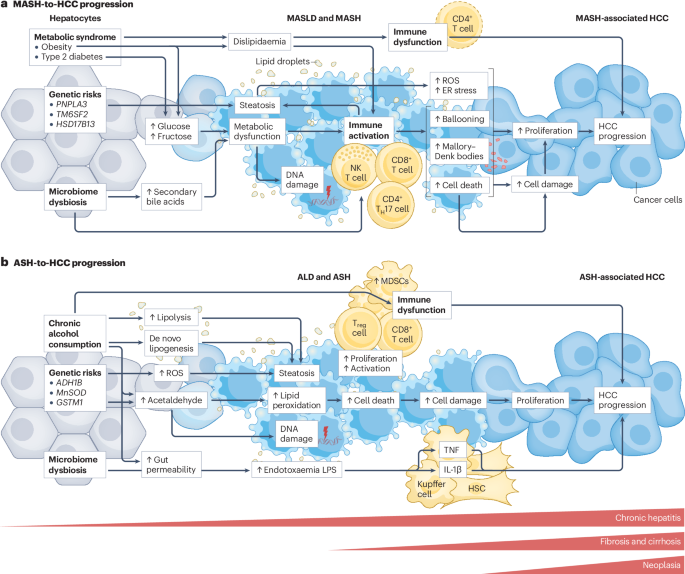
Steatotic liver diseases (SLDs), specifically metabolic dysfunction-associated steatotic liver disease (MASLD), are increasingly driving the global rise in hepatocellular carcinoma (HCC). By triggering chronic inflammation and metabolic dysfunction, these conditions accelerate liver cell mutation, making early screening and metabolic intervention critical for preventing liver cancer in non-viral patients.
For decades, the medical community viewed liver cancer primarily through the lens of viral hepatitis B and C. However, a tectonic shift is occurring. As global obesity and type 2 diabetes rates climb, the primary driver of liver malignancy is pivoting toward metabolic dysfunction. This is not merely a change in statistics; it is a change in the biological pathway to cancer. Unlike viral hepatitis, which often follows a predictable path of cirrhosis before malignancy, MASLD can trigger hepatocellular carcinoma (HCC) even in the absence of advanced scarring, creating a dangerous “silent” window for patients.
In Plain English: The Clinical Takeaway
- Fat is not just storage: Excess fat in the liver causes chronic inflammation, which acts like a “slow burn,” damaging DNA and increasing cancer risk.
- Cirrhosis isn’t always required: Unlike older beliefs, some people with metabolic liver disease can develop liver cancer without having severe scarring (cirrhosis) first.
- Prevention is metabolic: Managing blood sugar, weight, and lipids is now considered a primary frontline defense against liver cancer.
The Molecular Bridge: How Metabolic Stress Triggers Oncogenesis
The transition from a fatty liver to a malignant tumor involves a complex mechanism of action—the specific biochemical process through which a disease produces its effect. In MASLD, the liver becomes overwhelmed by free fatty acids, leading to lipotoxicity. This triggers oxidative stress, where unstable molecules called reactive oxygen species (ROS) damage cellular membranes and DNA.
This environment promotes a state of chronic, low-grade inflammation known as metabolic dysfunction-associated steatohepatitis (MASH). In this state, the immune system’s attempt to clear damaged cells actually releases pro-inflammatory cytokines, which can inadvertently signal liver cells to mutate and proliferate uncontrollably. This pathway is distinct from the direct cellular destruction seen in viral hepatitis, making the resulting tumors potentially more aggressive and harder to detect in early stages.
“The shift toward MASLD-driven liver cancer represents a public health crisis that mirrors the obesity epidemic. We are seeing a rise in HCC in patients who do not fit the traditional profile of a liver cancer patient, necessitating a complete overhaul of our screening protocols.” — Dr. Hepatologist and lead researcher in metabolic oncology.
Global Regulatory Landscapes and Patient Access
The clinical management of SLDs varies significantly across geographic borders. In the United States, the FDA has recently moved toward approving targeted therapies for MASH, such as Resmetirom, which targets the thyroid hormone receptor-beta to reduce liver fat and inflammation. This represents a pivot from “lifestyle-only” advice to pharmacological intervention.
In Europe, the EMA (European Medicines Agency) maintains a rigorous focus on long-term histological evidence—meaning they require proof that the actual structure of the liver has improved, not just that blood markers have dropped. Meanwhile, the NHS in the UK is integrating more non-invasive biomarkers, such as FibroScan, to identify high-risk patients without resorting to invasive biopsies. This disparity means that a patient’s ability to access “cancer-preventative” metabolic drugs depends heavily on their regional healthcare system’s willingness to fund high-cost biologics.
The research underpinning these shifts is largely funded by a combination of government grants (such as the NIH in the US) and pharmaceutical partnerships. While this accelerates drug development, it underscores the necessitate for double-blind placebo-controlled trials—studies where neither the patient nor the doctor knows who is receiving the treatment—to ensure that efficacy is not exaggerated by corporate bias.
Comparative Risk Profiles: Viral vs. Metabolic Liver Cancer
To understand the versatility of the interplay between SLDs and cancer, we must compare the risk trajectories of different liver disease etiologies.
| Feature | Viral Hepatitis (HBV/HCV) | MASLD/MASH | Combined (Dual Etiology) |
|---|---|---|---|
| Primary Driver | Viral Replication | Insulin Resistance/Lipids | Synergistic Inflammation |
| Path to HCC | Usually via Cirrhosis | Can bypass Cirrhosis | Accelerated Progression |
| Screening Standard | Every 6 Months (Ultrasound) | Risk-stratified (F3+ Fibrosis) | Aggressive Surveillance |
| Primary Intervention | Antivirals | Metabolic/GLP-1 Agonists | Combined Therapy |
The Role of GLP-1 Agonists in Cancer Prevention
A critical area of current investigation is the utilize of GLP-1 receptor agonists—originally designed for type 2 diabetes and weight loss—as a prophylactic measure against liver cancer. By improving systemic insulin sensitivity and reducing the hepatic fat fraction, these drugs may effectively “starve” the inflammatory environment that feeds early-stage tumors.
However, these are not without contraindications—specific situations in which a drug should not be used because it may be harmful. For instance, patients with a history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2 are generally advised against GLP-1 therapies. The medical community is currently debating whether the metabolic benefit of cancer prevention outweighs these specific risks in high-risk SLD populations.
Contraindications & When to Consult a Doctor
While metabolic improvements are generally beneficial, certain interventions require strict medical supervision. You should consult a hepatologist or gastroenterologist immediately if you experience:
- Jaundice: Yellowing of the skin or eyes, indicating liver dysfunction.
- Ascites: Rapid increase in abdominal girth or swelling.
- Unexplained Weight Loss: Combined with upper right quadrant abdominal pain.
- Elevated Liver Enzymes: If blood tests show persistently high ALT or AST levels.
Patients with severe renal impairment or a history of pancreatitis should exercise caution with latest metabolic medications and only use them under the guidance of a specialist.
The Future Trajectory of Liver Oncology
The interplay between steatotic liver disease and cancer suggests that we are moving toward a model of “precision hepatology.” Instead of treating all liver cancer the same, clinicians will soon tailor treatments based on whether the tumor was driven by a virus, a metabolic failure, or a combination of both. The goal is to move from reactive treatment (treating the tumor) to proactive prevention (treating the metabolic environment).
As we refine our understanding of the molecular triggers, the focus will shift toward early detection biomarkers that can spot a “pre-cancerous” metabolic liver long before a tumor appears on an ultrasound. The battle against liver cancer is no longer just about killing viruses; it is about managing the chemistry of the modern human diet.