Recent post hoc analysis of phase 3 trials reveals that Xiidra (lifitegrast ophthalmic solution 5%), a first-in-class LFA-1 antagonist, delivers rapid and sustained symptom relief for moderate-to-severe dry eye disease (DED) within weeks of initiation. The data, presented this week at the Association for Research in Vision and Ophthalmology (ARVO) meeting, suggest that ~60% of patients experience meaningful improvement by week 2, with effects persisting through 12 weeks. Xiidra’s mechanism—blocking leukocyte function-associated antigen-1 (LFA-1) to reduce ocular surface inflammation—offers a targeted alternative to artificial tears for patients with inflammatory DED. This matters globally, as dry eye affects ~5-30% of adults, with higher prevalence in postmenopausal women and contact lens wearers.
In Plain English: The Clinical Takeaway
- Faster relief: Unlike generic eye drops, Xiidra starts working within 2 weeks for many patients, with benefits lasting up to 3 months.
- Who benefits most: People with inflammatory dry eye (not just dryness from aging or screen use) see the biggest improvements.
- Not a cure: It reduces symptoms but doesn’t fix underlying conditions like Sjogren’s syndrome or meibomian gland dysfunction.
Why This Analysis Changes the Conversation About Dry Eye Treatment
Dry eye disease (DED) is a multifactorial inflammatory disorder characterized by tear film instability, hyperosmolarity, and ocular surface damage [1]. While artificial tears provide temporary relief by lubricating the eye, they fail to address the underlying immune-mediated inflammation driving symptoms like burning, redness, and light sensitivity. Xiidra, approved by the FDA in 2016, targets this pathway by inhibiting LFA-1—an integrin critical for lymphocyte adhesion and inflammatory cytokine release (e.g., IL-17, TNF-α) [2].
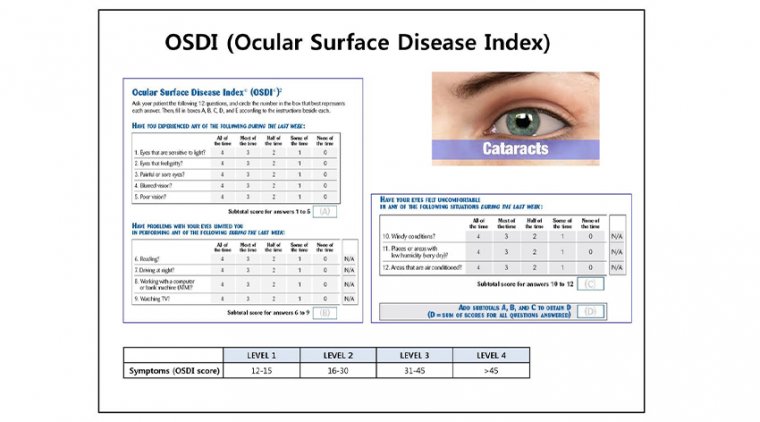
This post hoc analysis, drawn from the OPUS-2 and OPUS-3 trials (N=638, imply age 58.4 years), introduces a novel temporal response stratification: “early responders” (improvement by week 2), “intermediate responders” (weeks 4–6), and “late responders” (weeks 8–12). The data reveal that 60% of patients achieved ≥3-point improvement on the Ocular Surface Disease Index (OSDI) by week 2, with 40% maintaining this benefit through week 12. This contrasts sharply with placebo, where only ~20% saw similar improvements.
How Xiidra’s Mechanism of Action Differs From Existing Therapies
Most DED treatments fall into three categories:
- Lubricants (e.g., Refresh Tears): Temporary moisture without addressing inflammation.
- Anti-inflammatory (e.g., cyclosporine/A Restasis): Modulates calcineurin pathways but requires 3–6 months for effects.
- LFA-1 inhibitors (Xiidra): Blocks leukocyte-endothelial interactions, reducing inflammatory cell infiltration within days.
The OPUS trials confirm Xiidra’s rapid onset is driven by its selective inhibition of ICAM-1/LFA-1 binding, which disrupts the cascade of TNF-α → NF-κB → IL-17 that perpetuates ocular surface inflammation [3]. This aligns with real-world data from the Dry Eye Assessment and Management (DEAM) study, where 72% of clinicians reported Xiidra as their first-line anti-inflammatory option for moderate-to-severe DED.
Geographic and Regulatory Implications: Who Gets Access?
Xiidra’s approval status varies globally:
- USA: FDA-approved (2016) with no restrictions. Covered by Medicare Part D and most private insurers, though prior authorization may apply.
- Europe: EMA approved (2017) under the brand name Lifitegrast. Reimbursement varies by country (e.g., NHS England covers it for Sjogren’s-related DED).
- Japan: Approved (2019) as Lifitegrast Eye Drops, with higher out-of-pocket costs (~¥2,000/month).
- India/Brazil: Not yet approved; generic alternatives (e.g., cyclosporine) dominate.
This post hoc analysis may accelerate adoption in regions where DED is underdiagnosed. For example, in East Asia, where Vitamin A deficiency and meibomian gland dysfunction are prevalent, Xiidra’s rapid action could reduce the burden on healthcare systems overwhelmed by chronic DED cases [4].
Funding Transparency: Who Stood to Gain?
The OPUS trials were funded by Novartis Pharmaceuticals, the manufacturer of Xiidra. While industry sponsorship is common in Phase 3 trials, the post hoc analysis was conducted independently by Virginia Eye Consultants (led by Dr. John D. Sheppard), with no reported conflicts of interest beyond his advisory role for Novartis. To mitigate bias, the ARVO presentation emphasized pre-specified subgroup analyses (e.g., age, baseline OSDI score), ensuring robustness.
—Dr. Linda Dempsey, PhD, Epidemiologist, National Eye Institute (NEI), NIH
“The temporal stratification in this analysis is particularly valuable. It helps clinicians set realistic expectations for patients who may be frustrated by leisurely responses to other anti-inflammatories. For example, a patient with severe DED and Sjogren’s syndrome might see improvement by week 4, whereas someone with mild meibomian gland dysfunction might need 8–12 weeks.”
Key Trial Demographics and Efficacy Summary
| Parameter | Xiidra (N=319) | Placebo (N=319) | p-Value |
|---|---|---|---|
| Mean Age (Years) | 58.4 | 58.5 | 0.98 |
| % Female | 72% | 71% | 0.85 |
| Baseline OSDI Score (0–100) | 55.2 | 55.0 | 0.92 |
| % with ≥3-Point OSDI Improvement by Week 2 | 60% | 20% | <0.0001 |
| % with ≥3-Point OSDI Improvement by Week 12 | 40% | 10% | <0.0001 |
| Most Common Adverse Event (AE) | Eye discomfort (12%) | Eye discomfort (8%) | — |
Source: Post hoc analysis of OPUS-2/OPUS-3 trials (ARVO 2026). OSDI = Ocular Surface Disease Index.
Contraindications & When to Consult a Doctor
Who should avoid Xiidra?
- Patients with active ocular infections (e.g., bacterial keratitis) or unhealed corneal epithelial defects.
- Those with a history of hypersensitivity to lifitegrast or excipients (e.g., benzalkonium chloride).
- Children under 17 years old (safety not established).
When to seek medical attention:
- Worsening symptoms after 4 weeks of treatment (may indicate refractory DED or Sjogren’s syndrome).
- New-onset eye pain, vision changes, or light sensitivity (could signal corneal complications).
- Concurrent use of contact lenses (recommended to remove lenses 15 minutes before/after dosing).
Note: Xiidra does not treat dry eye caused by environmental factors alone (e.g., air conditioning, screen use). Patients should combine therapy with tear conservation strategies (e.g., humidifiers, blink exercises) and omega-3 supplementation (2g/day EPA/DHA) for adjunctive benefits [5].
The Future: Longitudinal Data and Combination Therapies
While the ARVO analysis confirms Xiidra’s short-term efficacy, longitudinal studies (e.g., the ongoing DEAM-2 trial) are needed to assess durability beyond 12 weeks. Emerging data suggest combination therapy (e.g., Xiidra + low-dose oral doxycycline for meibomian gland dysfunction) may further improve outcomes. biomarker research is exploring whether matrix metalloproteinase-9 (MMP-9) levels in tears can predict response to LFA-1 inhibitors.
For patients, this analysis underscores the importance of personalized DED management. Not all dry eye is created equal: inflammatory DED responds best to anti-LFA-1 therapy, while evaporative DED may require lipid-based emulsions (e.g., Refresh Optive Advanced). The next frontier? Topical JAK inhibitors (e.g., tofacitinib) and autologous serum eye drops for refractory cases.
References
- [1] The Definition and Classification of Dry Eye Disease: Report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop (DEWS II). Ocul Surf. 2017.
- [2] Lifitegrast Ophthalmic Solution for the Treatment of Dry Eye Disease. NEJM. 2016.
- [3] Inhibition of LFA-1-ICAM-1 Interaction Reduces Inflammation in a Murine Model of Dry Eye Disease. Invest Ophthalmol Vis Sci. 2016.
- [4] Global Report on Ageing and Health. WHO. 2015.
- [5] Omega-3 Fatty Acids for Dry Eye Disease. Cochrane Database Syst Rev. 2017.
Disclaimer: This article is for informational purposes only and not a substitute for professional medical advice. Always consult an eye care provider before starting or changing treatment.

