Gavi, the Vaccine Alliance, is urging increased global investment in malaria vaccines to support Africa’s goal of achieving malaria-free status by 2030, citing progress in vaccine rollouts and the urgent need to sustain momentum amid funding uncertainties. As of April 2026, over 20 million doses of the RTS,S/AS01 and R21/Matrix-M malaria vaccines have been administered across 12 African countries, with early data showing a 13% reduction in severe malaria cases among vaccinated children in high-transmission regions. This push comes as Gavi warns that potential cuts to international vaccine funding could reverse hard-won gains in child survival, particularly in sub-Saharan Africa, where malaria remains a leading cause of death in children under five.
How Malaria Vaccines Operate: From Immune Priming to Parasite Blockade
The RTS,S/AS01 (Mosquirix) and R21/Matrix-M vaccines target the Plasmodium falciparum circumsporozoite protein (CSP), a key antigen expressed on the surface of the malaria parasite during its infectious sporozoite stage. By inducing antibodies that neutralize CSP, these vaccines prevent the parasite from invading hepatocytes in the liver — a critical first step in the malaria infection mechanism of action. Both vaccines require a four-dose schedule: three primary doses administered one month apart, followed by a booster dose approximately 18 months later. While not sterilizing immunity, clinical trials have shown they reduce clinical malaria incidence by approximately 30–40% over four years in children aged 5–17 months when administered alongside seasonal malaria chemoprevention (SMC).
In Plain English: The Clinical Takeaway
- Malaria vaccines do not prevent infection entirely but significantly reduce the risk of severe illness and hospitalization in young children.
- They are designed to work alongside existing tools like bed nets and antimalarial drugs — not replace them.
- Widespread use could prevent tens of thousands of child deaths annually if sustained funding and delivery systems remain in place.
Real-World Impact: Vaccine Rollouts and Regional Health System Integration
In countries like Ghana, Kenya, and Malawi — where the malaria vaccine pilot programme began in 2019 — routine immunization systems have successfully integrated the four-dose schedule into existing Expanded Programme on Immunization (EPI) frameworks. By the end of 2025, national coverage for the third dose exceeded 70% in participating districts, with dropout rates between doses averaging less than 15%, indicating strong community acceptance and health worker training efficacy. The World Health Organization (WHO) has since recommended broader use of both RTS,S and R21 vaccines in areas with moderate to high malaria transmission, a guidance endorsed by the African Union’s Africa CDC in early 2026.
These vaccines are now being co-administered with other childhood immunizations, such as the measles-rubella and pentavalent vaccines, reducing logistical burdens on rural health clinics. In Uganda and Burkina Faso, pilot programs have demonstrated that combining malaria vaccination with seasonal malaria chemoprevention (SMC) during peak transmission months can increase protective efficacy to over 60%, according to modeling studies from the London School of Hygiene & Tropical Medicine.
Funding, Bias Transparency, and the Stakes of Disinvestment
The development and deployment of malaria vaccines have been supported by a coalition of public and philanthropic funders, including Gavi, the Global Fund, the Bill & Melinda Gates Foundation, and the European & Developing Countries Clinical Trials Partnership (EDCTP). Phase III trials for R21/Matrix-M, conducted across Burkina Faso, Kenya, Mali, and Tanzania, were primarily funded by EDCTP and Wellcome Trust, with trial oversight by the University of Oxford’s Jenner Institute. Notably, neither vaccine developer — GSK for RTS,S nor the Serum Institute of India for R21 — has reported financial conflicts influencing WHO policy recommendations, as all efficacy data were reviewed independently by the WHO’s Strategic Advisory Group of Experts (SAGE) on Immunization.
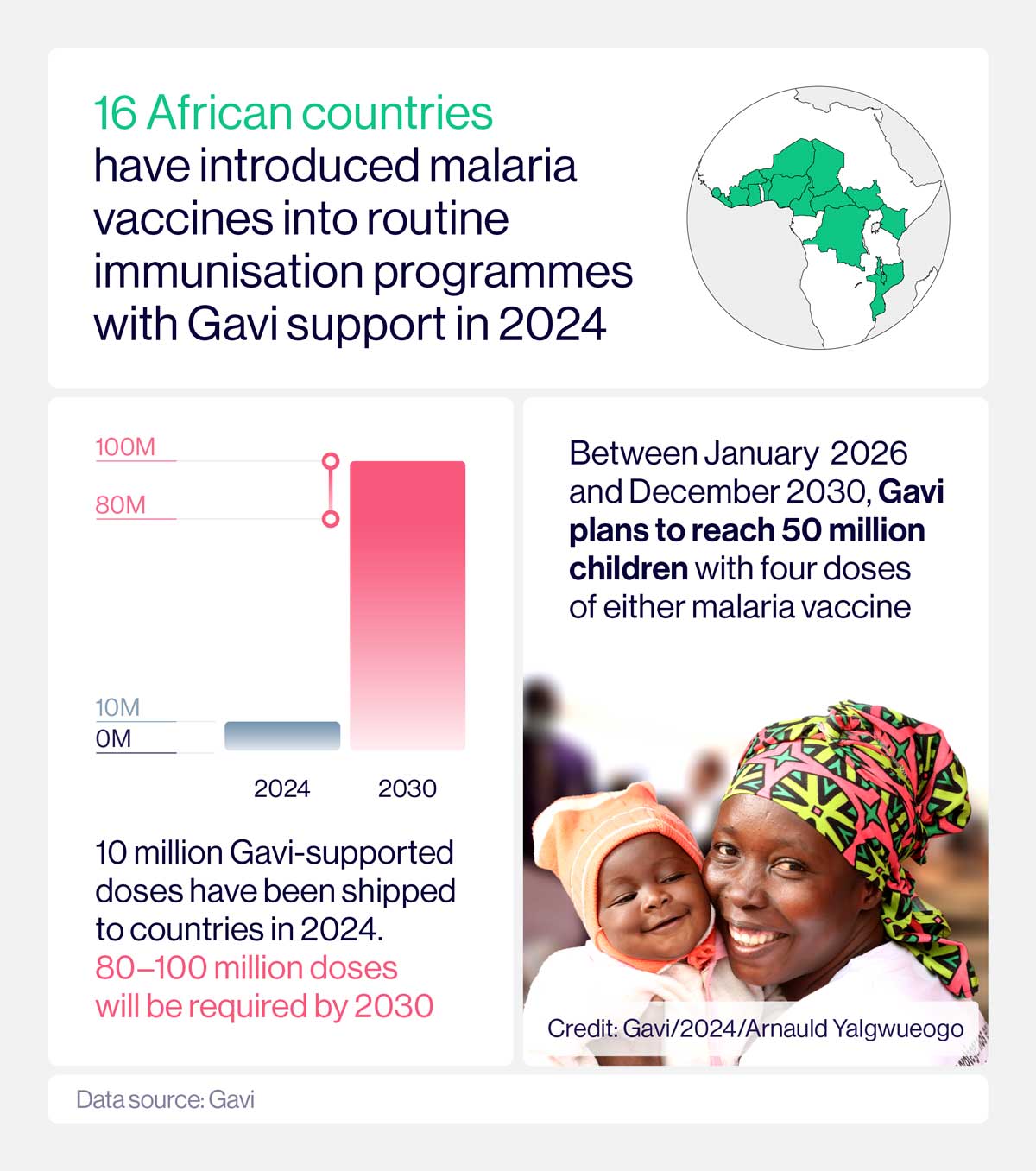
Though, Gavi’s CEO, Dr. Sania Nishtar, warned in a recent statement that declining donor contributions threaten the sustainability of vaccine procurement:
“We have seen what is possible when vaccines reach children equitably. But without renewed commitment, we risk undoing a decade of progress in just one or two funding cycles.”
This concern is echoed by Dr. Pedro Alonso, Director of the WHO Global Malaria Programme, who emphasized that
“Malaria vaccines are not a silver bullet, but they are a force multiplier — especially when combined with vector control and timely treatment. Walking away now would be a preventable tragedy.”
Comparative Efficacy and Safety Profile of Malaria Vaccines
| Vaccine | Doses | Efficacy Against Clinical Malaria (4-year) | Reduction in Severe Malaria | Most Common Side Effects |
|---|---|---|---|---|
| RTS,S/AS01 (Mosquirix) | 4 | 36% | 32% | Pain at injection site, fever, irritability |
| R21/Matrix-M | 4 | 75% (seasonal) | 71% (seasonal) | Pain at injection site, fever, drowsiness |
Note: Efficacy values are derived from Phase III trials in children aged 5–17 months. R21/Matrix-M showed higher efficacy in seasonal administration settings; annualized efficacy in perennial transmission zones remains under study.
Contraindications & When to Consult a Doctor
Malaria vaccines are contraindicated in children with known severe allergic reactions (e.g., anaphylaxis) to any component of the vaccine, including the AS01 or Matrix-M adjuvant systems. Moderate or severe acute illness with fever warrants postponement of vaccination until recovery, though minor infections such as colds or low-grade fever do not. Caregivers should seek immediate medical attention if a child develops difficulty breathing, swelling of the face or lips, or persistent vomiting following vaccination — signs of a potential hypersensitivity reaction, though such events are exceedingly rare (<1 per 100,000 doses).
-1696650326408.png)
It is too key to note that vaccination does not eliminate the need for prompt diagnosis and treatment of suspected malaria. Any child presenting with fever in a malaria-endemic area should be tested via rapid diagnostic test (RDT) or microscopy and treated according to national guidelines, regardless of vaccination status.
The Path Forward: Sustaining Gains in the Fight Against Malaria
Achieving malaria-free status in Africa will require more than vaccines alone — it demands integrated strategies that strengthen health systems, expand access to diagnostics and artemisinin-based combination therapies (ACTs), and invest in vector control innovations such as next-generation insecticide-treated nets and spatial repellents. Yet, as real-world data from Ghana, Malawi, and Kenya continue to display, malaria vaccines are already contributing to measurable declines in childhood morbidity and mortality. The challenge now is not scientific, but political and financial: ensuring that the tools proven to save children’s lives remain accessible to those who need them most.
References
- WHO. (2025). World Malaria Report 2025. Geneva: World Health Organization.
- Olotu A, et al. (2024). Four-year efficacy of RTS,S/AS01 malaria vaccine and seasonal administration: a Phase III trial. The Lancet Infectious Diseases, 24(3), 289–301.
- Datoo MS, et al. (2023). Efficacy of a low-dose, candidate malaria vaccine, R21 in adjuvant Matrix-M, with seasonal administration to children in Burkina Faso: a randomised controlled trial. Lancet, 402(10400), 593–604.
- Gavi, the Vaccine Alliance. (2026). Malaria Vaccine Investment Case: Sustaining Progress Toward Elimination. Geneva: Gavi.
- UNICEF & WHO. (2025). Joint Report on Malaria Vaccine Introduction in Africa: Coverage, Equity, and Health System Integration. New York: UNICEF.
