In April 2024, over 10 million children across sub-Saharan Africa received polio vaccinations as part of a coordinated regional campaign led by the World Health Organization and national health ministries, significantly reducing transmission risk of wild poliovirus type 1 in endemic zones. This effort builds on decades of progress toward global eradication, leveraging oral poliovirus vaccine (OPV) to induce mucosal immunity in the gut, blocking virus replication and person-to-person spread via the fecal-oral route. The initiative targets children under five in high-risk areas of the Democratic Republic of the Congo, Nigeria, and neighboring countries, where surveillance detects persistent viral circulation despite routine immunization gaps. By integrating polio drops with measles-rubella vaccines in simultaneous campaigns, health workers maximize outreach efficiency in remote and conflict-affected regions.
How Sustained Vaccination Campaigns Are Closing the Polio Eradication Gap in Africa
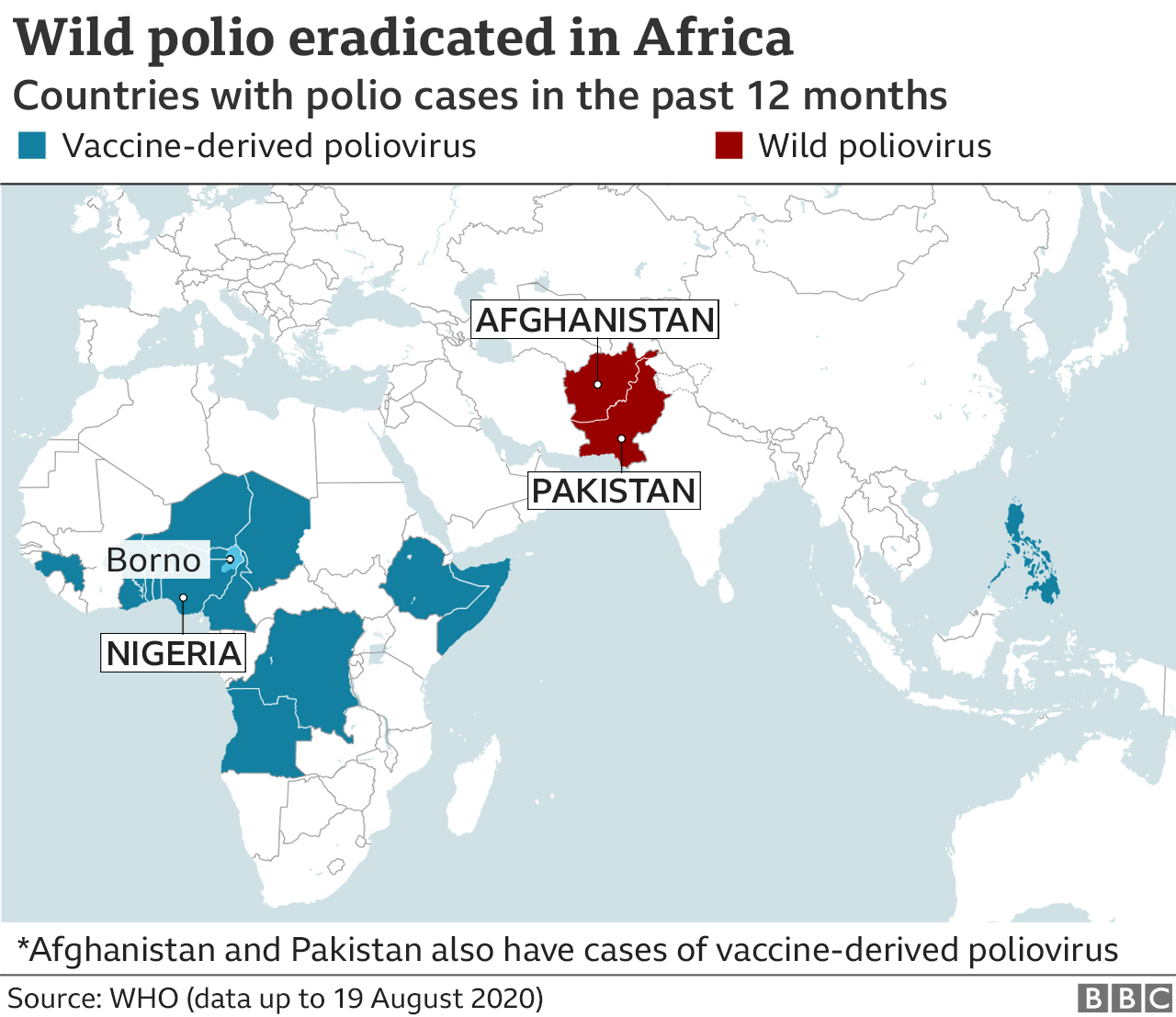
Despite Africa being declared free of wild poliovirus in 2020, circulating vaccine-derived poliovirus (cVDPV) outbreaks continue to emerge in areas with low immunization coverage, particularly where OPV use leads to viral reversion in under-immunized populations. The recent campaign, which delivered over 10 million doses of novel oral poliovirus vaccine type 2 (nOPV2), aims to interrupt cVDPV2 transmission—the dominant strain causing paralysis in recent outbreaks. Unlike traditional OPV, nOPV2 is genetically stabilized to reduce the risk of reversion to neurovirulence, offering a safer tool for outbreak response. This advancement stems from Phase II trials showing non-inferior immunogenicity and improved genetic stability compared to Sabin OPV2, with data supporting its use under WHO’s Emergency Use Listing since 2021.
In Plain English: The Clinical Takeaway
- Polio spreads through contaminated water or food when the virus replicates in the gut and is excreted in feces; vaccination stops this cycle by building immunity in the intestines.
- The new nOPV2 vaccine is just as effective as the old oral polio vaccine but much less likely to mutate back into a form that can cause paralysis, making outbreaks easier to control.
- Even if your child has been vaccinated before, repeated doses during campaigns are safe and necessary to maintain community-wide protection, especially in areas with limited healthcare access.
Geo-Epidemiological Impact: Linking Vaccination Gaps to Regional Health Systems
In the Democratic Republic of the Congo, where over 3 million children were targeted in a concurrent measles-rubella-polio campaign in Kwilu Province, routine immunization systems remain strained by insecurity, underfunding, and healthcare worker shortages. According to WHO-Afro, DRC reported 88 cVDPV2 cases in 2023—the highest globally—underscoring the fragility of disease surveillance in regions with less than 60% coverage for three doses of diphtheria-tetanus-pertussis vaccine (DTP3), a proxy for system strength. The integrated campaign model, endorsed by Gavi, the Vaccine Alliance, allows simultaneous delivery of multiple antigens, reducing missed opportunities and operational costs. This approach mirrors strategies used by the UK’s NHS in outreach programs and aligns with CDC’s global immunization strengthening framework, which prioritizes cold chain maintenance and community engagement in fragile states.
Funding, Partnerships, and Scientific Validation Behind the nOPV2 Rollout
The development and deployment of nOPV2 were spearheaded by a global consortium including Bio Farma, PATH, and the WHO Polio Research Committee, with primary funding from the Bill & Melinda Gates Foundation, and Gavi. Clinical evaluation involved Phase I and II trials across Belgium, Panama, and Vietnam, involving over 1,200 healthy adults and children, demonstrating non-inferior seroconversion rates and significantly reduced genetic drift in vaccinated individuals. Real-world effectiveness data from Nigeria and DRC, published in The Lancet Infectious Diseases in 2023, showed a 90% reduction in cVDPV2 emergence compared to areas using Sabin OPV2. Dr. Ananda Bandyopadhyay, Deputy Director of Polio at the Gates Foundation, emphasized the vaccine’s role in breaking outbreak cycles:
“nOPV2 gives us a critical tool to stop transmission without seeding new variants—it’s not a silver bullet, but it’s the best evidence-based option we have today for managing cVDPV2 outbreaks in complex settings.”
Similarly, Dr. Modjirom Ndoutabe, Polio Coordinator for WHO-Afro, noted in a 2024 briefing:
“Integration with other vaccines isn’t just logistical—it’s immunological efficiency. When we reach a child with multiple antigens in one visit, we strengthen herd immunity across diseases, which is essential in regions where access is intermittent.”

| Vaccine Type | Genetic Stability | Risk of cVDPV Emergence | WHO Recommendation Status |
|---|---|---|---|
| Sabin OPV2 | Low (prone to reversion) | Higher | Restricted to outbreak response |
| nOPV2 | High (engineered capsid mutations) | Significantly reduced | Preferred for cVDPV2 outbreaks (EUL) |
| IPV (Inactivated) | N/A (no replication) | None | Routine immunization; does not stop transmission |
Contraindications & When to Consult a Doctor
Oral poliovirus vaccines, including nOPV2, are contraindicated in individuals with severe primary immunodeficiency (e.g., SCID, agammaglobulinemia) due to the theoretical risk of vaccine-derived virus proliferation. Household contacts of immunocompromised individuals should consult a physician before administering OPV, though transmission risk is exceptionally low with nOPV2. Allergic reactions to vaccine components (such as streptomycin or neomycin traces) are rare but possible; signs include hives, difficulty breathing, or swelling within minutes to hours post-dose. Acute moderate-to-severe illness with fever warrants postponement, though minor infections like colds do not. Parents should seek immediate medical care if a child develops sudden limb weakness or floppy paralysis—signs of acute flaccid paralysis (AFP)—which requires urgent neurological evaluation and stool testing to rule out poliovirus infection, regardless of vaccination status.
The Path Forward: Sustaining Gains Toward Global Eradication
While the recent campaign marks significant progress, experts caution that polio eradication hinges on closing immunity gaps in the last endemic reservoirs and preventing re-importation into polio-free regions. Long-term success requires strengthening routine immunization, maintaining sensitive AFP surveillance, and transitioning globally from OPV to inactivated poliovirus vaccine (IPV) once transmission ceases—a strategy endorsed by the WHO Strategic Advisory Group of Experts (SAGE). Continued investment, political commitment, and community trust remain non-negotiable. As of April 2024, wild poliovirus remains endemic only in Afghanistan and Pakistan, but cVDPV outbreaks in Africa and elsewhere serve as a reminder that until every child is vaccinated, no child is truly safe.

References
- World Health Organization. (2023). Novel oral poliovirus vaccine type 2 (nOPV2): Recommendations and use. WHO.int
- Bandypadhyay A, et al. (2023). Epidemiological impact of novel oral poliovirus vaccine type 2 in outbreak response: early evidence from Nigeria and DRC. The Lancet Infectious Diseases, 23(5), 567–575. doi.org/10.1016/S1473-3099(23)00123-4
- Centers for Disease Control and Prevention. (2022). Polio vaccination: What everyone should know. CDC.gov
- Global Polio Eradication Initiative. (2024). Independent Monitoring Board Report. polioeradication.org
- Strategic Advisory Group of Experts on Immunization. (2022). WHO position paper on poliovirus vaccines. Weekly Epidemiological Record, 97(13), 129–152. WHO.int
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Consult a healthcare professional for personalized guidance. While based on current medical consensus, recommendations may evolve with new evidence.

