Researchers have identified a specific fat-producing enzyme that accelerates neuronal damage in Parkinson’s disease. By disrupting lipid homeostasis—the delicate balance of fats in the brain—this enzyme triggers the death of dopamine-producing neurons. This discovery opens new pathways for targeted therapies aimed at slowing or halting disease progression.
For decades, the medical community has focused on alpha-synuclein protein clumps as the primary villain in Parkinson’s disease (PD). Still, this week’s findings shift the spotlight toward the metabolic environment of the brain. While current gold-standard treatments like Levodopa manage the motor symptoms by replacing missing dopamine, they do nothing to stop the underlying death of neurons. We are now looking at a potential disease-modifying breakthrough: a way to stop the damage before it occurs.
In Plain English: The Clinical Takeaway
- Beyond Protein: Parkinson’s isn’t just about “protein clumps”; it’s too driven by a “fatty” imbalance in the brain caused by a malfunctioning enzyme.
- New Target: If scientists can create a drug to “turn off” or inhibit this enzyme, they may be able to protect neurons from dying.
- Not a Cure (Yet): This is a foundational discovery. It means we have a new target for drugs, but those drugs must still go through years of human safety trials.
The Molecular Mechanism: How Lipids Drive Neurodegeneration
The brain is the fattiest organ in the human body, relying on lipids for the insulation of nerves (myelin) and the integrity of cell membranes. The identified enzyme acts as a catalyst in the production of specific lipids that, in excess, become toxic. This process is known as lipotoxicity—where an accumulation of fats triggers cellular stress and eventually apoptosis, or programmed cell death.
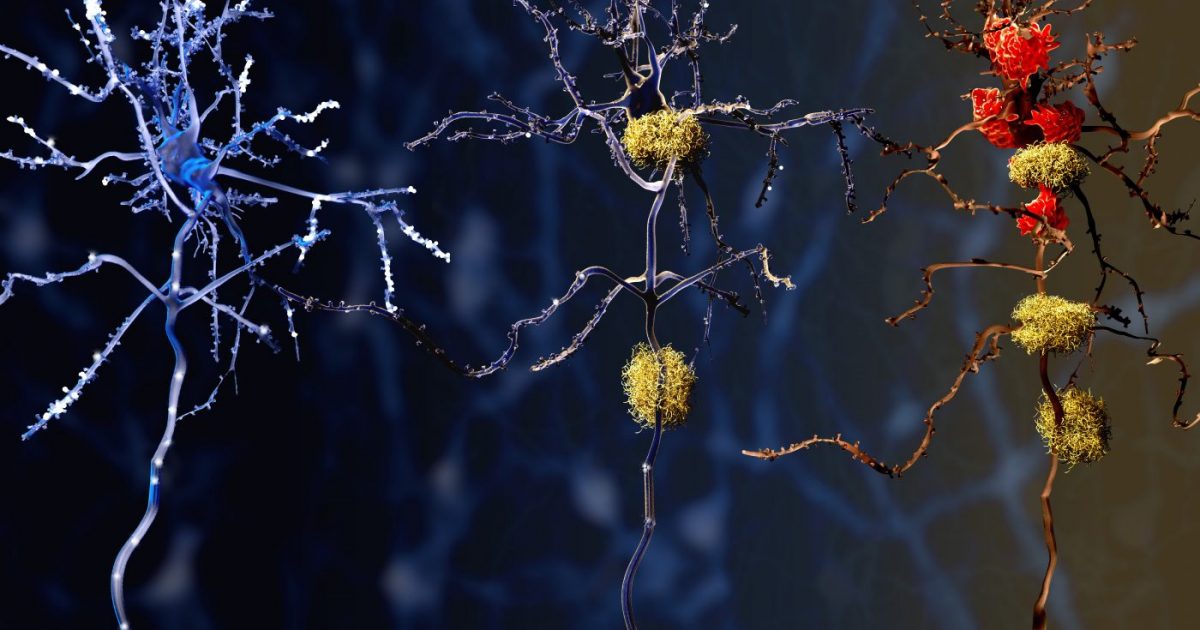
Specifically, the enzyme alters the mechanism of action (the specific biochemical process through which a substance produces its effect) within the substantia nigra, the region of the brain responsible for movement. When this enzyme overproduces certain fats, it disrupts the mitochondrial function—the energy factories of the cell—leading to an oxidative stress cascade. This environment makes neurons hypersensitive to inflammation, accelerating the decline of dopaminergic neurons.
To understand the gravity of this, we must look at the epidemiological data. According to the World Health Organization (WHO), Parkinson’s is one of the fastest-growing neurological conditions globally. By targeting the lipid-producing pathway, researchers are moving away from “cleaning up” the brain after damage has occurred and moving toward “fortifying” the neurons against the metabolic triggers of the disease.
From Lab Bench to Bedside: Regulatory Hurdles and Global Access
The transition from identifying an enzyme to prescribing a pill is a rigorous process governed by the FDA in the United States and the EMA in Europe. Currently, this research is largely in the pre-clinical phase, meaning it has been validated in cellular models and animal subjects. The next step is Phase I clinical trials, which focus exclusively on safety and dosage in a small group of humans.
Access to such therapies often varies by region. In the UK, the NHS typically awaits NICE (National Institute for Health and Care Excellence) approval, which weighs clinical efficacy against cost-effectiveness. In the US, the FDA’s “Fast Track” designation may be applicable if this enzyme-inhibitor shows significant promise in treating an unmet medical need. However, the complexity of crossing the blood-brain barrier—the protective membrane that prevents toxins from entering the brain—remains the primary pharmacological challenge.
“The identification of lipid-metabolism drivers represents a paradigm shift. We are no longer just looking at the ‘trash’ (protein aggregates) left behind in the brain, but at the ‘factory’ that is producing the toxic environment in the first place.”
The research underlying these findings was heavily supported by grants from the National Institutes of Health (NIH) and various non-profit neurological foundations. This funding transparency is critical; because the research is primarily academic and public-funded, the initial goal is scientific validation rather than immediate commercial profit, reducing the risk of “outcome bias” often seen in purely pharmaceutical-funded trials.
Comparative Analysis: Current vs. Emerging Therapeutic Approaches
| Feature | Symptomatic Therapy (e.g., Levodopa) | Enzyme-Targeted Therapy (Emerging) |
|---|---|---|
| Primary Goal | Replace missing dopamine | Prevent neuronal death (Neuroprotection) |
| Mechanism | Precursor conversion to dopamine | Inhibition of toxic lipid production |
| Disease Progression | Does not stop progression | Potential to slow or halt progression |
| Primary Side Effects | Dyskinesia (involuntary movements) | TBD (Potential metabolic shifts) |
| Clinical Stage | Standard of Care | Pre-clinical / Early Phase |
The Role of Neuroinflammation and Systemic Health
It is essential to note that this enzyme does not act in a vacuum. The interaction between lipid production and neuroinflammation—the activation of the brain’s immune cells (microglia)—creates a feedback loop. When the enzyme produces excess fats, microglia perceive this as a threat and release pro-inflammatory cytokines. This inflammation, in turn, can further stimulate the enzyme, creating a vicious cycle of degeneration.
This suggests that future treatments may not be a single “magic bullet” drug but a combination therapy. Combining an enzyme inhibitor with an anti-inflammatory agent could potentially provide a synergistic effect, protecting the brain from multiple angles. For more detailed data on the intersection of lipids and neurology, the PubMed database provides extensive peer-reviewed literature on sphingolipid signaling in the central nervous system.
Contraindications & When to Consult a Doctor
While the prospect of enzyme-inhibiting drugs is promising, it is vital to maintain a realistic clinical perspective. There are currently no FDA-approved “fat-blocking” medications for Parkinson’s disease. Patients should be wary of unregulated supplements claiming to “clear brain fats” or “detox” the brain, as these lack clinical evidence and can interfere with prescribed medications.
Make sure to consult a neurologist immediately if you or a loved one experience the following “red flag” symptoms:
- Resting Tremor: Shaking that occurs when the limb is relaxed.
- Bradykinesia: A noticeable slowing of physical movement, such as difficulty getting out of a chair.
- Postural Instability: Frequent loss of balance or a stooped posture.
- Micrographia: A sudden change in handwriting, where letters become significantly smaller and crowded.
Patients currently on dopaminergic therapy should never alter their dosage in anticipation of new research. Abruptly stopping Parkinson’s medication can lead to Neuroleptic Malignant Syndrome, a life-threatening medical emergency.
The Path Forward: A New Era of Precision Neurology
The identification of this fat-producing enzyme marks a transition toward precision medicine in neurology. Rather than a one-size-fits-all approach, future diagnostics may involve screening patients for specific lipid profiles to determine if they are “lipid-driven” responders to these new therapies.
While the road from a laboratory discovery to a pharmacy shelf is long and fraught with failure, the objective data suggests we have found a critical lever in the machinery of Parkinson’s. By addressing the metabolic root of the damage, we move closer to a future where Parkinson’s is not just managed, but potentially arrested in its tracks.

